Advertisements
Advertisements
प्रश्न
Write a short note on Catenation.
उत्तर
Catenation: The property of carbon element due to which its atoms can join one another to form long carbon chains is called catenation.
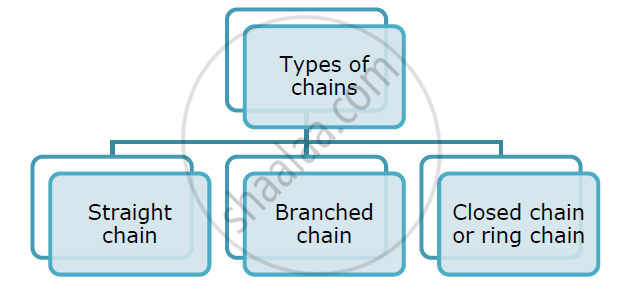
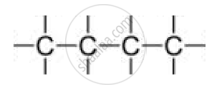
A) Straight chain of carbon atoms
B) Branched-chain of carbon atoms

C) Closed chain or ring chain of carbon atoms

APPEARS IN
संबंधित प्रश्न
Give any two differences between alkanes and alkenes.
Why does carbon form strong bonds with most other elements ?
What are the two properties of carbon which lead to the huge number of carbon compounds we see around us?
Draw the electron dot structures for propanone.
Give a test that can be used to differentiate chemically between butter and cooking oil.
Write the electron-dot structures for ethyne.
A cyclic hydrocarbon having carbon-carbon single bonds as well as carbon-carbon double bonds in its molecule is:
(a) C6H12
(b) C6H14
(c) C6H6
(d) C6H10
Write the molecular formula of an alkene having 20 carbon atoms?
A mixture of ethyne (acetylene) and oxygen is burnt for welding. Can you tell why a mixture of ethyne and air is not used?
Which of the following hydrocarbons can decolourise bromine water and which cannot? Why?
C6H12, C6H14, C6H10
Structural formula of ethyne is ______.
Identify the unsaturated compounds from the following:
- Propane
- Propene
- Propyne
- Chloropropane
Identify the unsaturated compounds from the following
- Propane
- Propene
- Propyne
- Chloropropane
Pentane has the molecular formula C5 H12 It has
Which among the following are unsaturated hydrocarbons?
- \[\ce{H3C - CH2 - CH2 - CH3}\]
- \[\ce{H3C - C ≡ C - CH3}\]
- \[\begin{array}{cc}
\ce{H3C - CH - CH3}\\
|\phantom{..}\\
\ce{CH3}\\
\end{array}\] - \[\begin{array}{cc}
\ce{H3C - C = CH2}\\
|\\
\phantom{...}\ce{CH3}\\
\end{array}\]
Write the name of the following compound
\[\begin{array}{cc}
\phantom{....}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{O}\phantom{......}\\
\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}||\phantom{......}\\
\ce{H} - \ce{C} - \ce{C} - \ce{C} - \ce{C} - \ce{C} - \ce{OH}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{.......}\\
\phantom{}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{.......}\\
\end{array}\]
Name the reaction which is commonly used in the conversion of vegetable oils to fats. Explain the reaction involved in detail.
Write the name of the following compound
\[\begin{array}{cc}
\phantom{}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{}\\
\ce{H - C - C - C - C - C - C - C = O}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{.....}\\
\phantom{.}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{......}
\end{array}\]
