Advertisements
Advertisements
प्रश्न
Draw the electron dot structures for ethanoic acid.
उत्तर
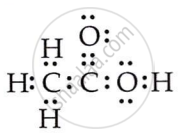
OR
\[\begin{array}{cc}
\phantom{.}\ce{H}\phantom{...}\ce{O}\phantom{.....}\\
\phantom{..}|\phantom{....}||\phantom{......}\\
\ce{H - C - C - O - H}\\
|\phantom{.........}\\
\ce{H}\phantom{.........}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
Draw the electron dot structures for propanone.
Give a test that can be used to differentiate chemically between butter and cooking oil.
A compound 'X' on heating with excess conc. sulphuric acid at 443 K gives an unsaturated compound 'Y'. 'X' also reacts with sodium metal to evolve a colourless gas 'Z'. Identify 'X', 'Y' and 'Z'. Write the equation of the chemical reaction of formation of 'Y' and also write the role of sulphuric acid in the reaction.
Write the electron-dot structures for ethyne.
Give the names and structural formulae of one saturated cyclic hydrocarbon and one unsaturated cyclic hydrocarbon.
Out of the following pairs of compounds, the unsaturated compounds are:
(a) C2H6 and C4H6
(b) C6H12 and C5H12
(c) C4H6 and C6H12
(d) C2H6 and C4H10
Which of the following will give addition reaction and why?
C4H10, C2H6, C2H4, CH4, C3H8, C3H4
Name a chemical reaction which is characteristic of unsaturated hydrocarbons (like alkenes and alkynes).
Name the reaction which is usually used in the conversion of vegetables oils to fats. Explain the reaction involved in detail. Write a chemical equation to illustrate your answer.
Identify the unsaturated compounds from the following:
- Propane
- Propene
- Propyne
- Chloropropane
Identify the unsaturated compounds from the following
- Propane
- Propene
- Propyne
- Chloropropane
Which among the following are unsaturated hydrocarbons?
- \[\ce{H3C - CH2 - CH2 - CH3}\]
- \[\ce{H3C - C ≡ C - CH3}\]
- \[\begin{array}{cc}
\ce{H3C - CH - CH3}\\
|\phantom{..}\\
\ce{CH3}\\
\end{array}\] - \[\begin{array}{cc}
\ce{H3C - C = CH2}\\
|\\
\phantom{...}\ce{CH3}\\
\end{array}\]
The name of the compound CH3 — CH2 — CHO is
Write the name of the following compound
\[\begin{array}{cc}
\phantom{....}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{O}\phantom{......}\\
\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}||\phantom{......}\\
\ce{H} - \ce{C} - \ce{C} - \ce{C} - \ce{C} - \ce{C} - \ce{OH}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{.......}\\
\phantom{}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{.......}\\
\end{array}\]
Name the functional groups present in the following compounds
- CH3 CO CH2 CH2 CH2 CH3
- CH3 CH2 CH2 COOH
- CH3 CH2 CH2 CH2 CHO
- CH3 CH2 OH
What is the role of metal or reagents written on arrows in the given chemical reactions?
- \[\begin{array}{cc}
\ce{CH3}\phantom{.....}\ce{CH3}\phantom{.........}\ce{CH3}\phantom{..}\ce{CH3}\phantom{..........}\\
\phantom{...}\backslash\phantom{......}/\phantom{................}|\phantom{.....}|\phantom{............}\\
\ce{C} = \ce{C} + \ce{H2} \overset{\ce{Ni}}{\rightarrow} \ce{CH3 - C - C - CH3}\\
\phantom{.}/\phantom{........}\backslash\phantom{..............}|\phantom{....}|\phantom{.........}\\
\ce{CH3}\phantom{....}\ce{CH3}\phantom{............}\ce{H}\phantom{...}\ce{H}\phantom{........}
\end{array}\] - \[\ce{CH3COOH + CH3CH2OH \overset{\ce{Conc. H2SO4}}{\rightarrow}CH3COOC2H5 + H2O}\]
- \[\ce{CH3CH2OH}\ce{->[Alk.KMnO4][Heat]CH3COOH}\]
Draw the electron dot structure for ethyne.
