Advertisements
Advertisements
Question
Draw the electron dot structures for ethanoic acid.
Solution
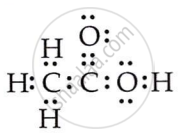
OR
\[\begin{array}{cc}
\phantom{.}\ce{H}\phantom{...}\ce{O}\phantom{.....}\\
\phantom{..}|\phantom{....}||\phantom{......}\\
\ce{H - C - C - O - H}\\
|\phantom{.........}\\
\ce{H}\phantom{.........}
\end{array}\]
APPEARS IN
RELATED QUESTIONS
Give a chemical test to distinguish between saturated and unsaturated hydrocarbons.
Why does carbon form strong bonds with most other elements ?
Draw the electron dot structures for H2S.
Give the names and structural formulae of one saturated cyclic hydrocarbon and one unsaturated cyclic hydrocarbon.
How many isomers of the following hydrocharbons are possible?
C3H8
An unsaturated hydrocarbon having a triple covalent bond has 50 hydrogen atoms in its molecule. The number of carbon atoms in its molecule will be:
(a) 24
(b) 25
(c) 26
(d) 28
Which of the following will give addition reaction and why?
C4H10, C2H6, C2H4, CH4, C3H8, C3H4
What happens when vegetable oils are hydrogenated? Name the catalyst used.
Which among the following are unsaturated hydrocarbons?
- \[\ce{H3C - CH2 - CH2 - CH3}\]
- \[\ce{H3C - C ≡ C - CH3}\]
- \[\begin{array}{cc}
\ce{H3C - CH - CH3}\\
|\phantom{..}\\
\phantom{.}\ce{CH3}
\end{array}\] - \[\begin{array}{cc}
\ce{H3C - C = CH2}\\
|\phantom{.}\\
\phantom{..}\ce{CH3}
\end{array}\]
Structural formula of ethyne is ______.
Structural formula of ethyne is
Carbon, Group (14) element in the Periodic Table, is known to form compounds with many elements. Write an example of a compound formed with
- chlorine (Group 17 of Periodic Table)
- oxygen (Group 16 of Periodic Table)
A compound X is formed by the reaction of a carboxylic acid C2H4O2 and an alcohol in presence of a few drops of H2SO4. The alcohol on oxidation with alkaline KMnO4 followed by acidification gives the same carboxylic acid as used in this reaction. Give the names and structures of
- carboxylic acid
- alcohol and
- the compound X.
Also write the reaction.
- What are hydrocarbons? Give examples.
- Give the structural differences between saturated and unsaturated hydrocarbons with two examples each.
- What is a functional group? Give examples of four different functional groups.
Write the name of the following compound
\[\begin{array}{cc}
\phantom{}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{}\\
\ce{H - C - C - C - C - C - C - C = O}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{.....}\\
\phantom{.}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{......}
\end{array}\]
