Advertisements
Advertisements
Question
Structural formula of ethyne is
Options
`"H" - "C" ≡ "C "- "H"`
`"H"_3-"C" ≡ "C" - "H"`
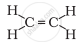
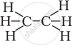
Solution
`"H" - "C" ≡ "C "- "H"`
The formula of ethyne is C2H2. There is one triple bond between carbon atoms.
APPEARS IN
RELATED QUESTIONS
What are the two properties of carbon which lead to the huge number of carbon compounds we see around us?
Giving their structures, state the number of single bonds, double bonds and triple bonds (if any) in the following compounds:
ethyne
Out of the following pairs of compounds, the unsaturated compounds are:
(a) C2H6 and C4H6
(b) C6H12 and C5H12
(c) C4H6 and C6H12
(d) C2H6 and C4H10
Which of the following hydrocarbons can decolourise bromine water and which cannot? Why?
C6H12, C6H14, C6H10
Name a cyclic unsaturated carbon compound.
How many double bonds are there in a saturated hydrocarbon?
Carbon, Group (14) element in the Periodic Table, is known to form compounds with many elements. Write an example of a compound formed with
- chlorine (Group 17 of Periodic Table)
- oxygen (Group 16 of Periodic Table)
Name the reaction which is commonly used in the conversion of vegetable oils to fats. Explain the reaction involved in detail.
Write the name of the following compound
\[\begin{array}{cc}
\phantom{}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{}\\
\ce{H - C - C - C - C - C - C - C = O}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{.....}\\
\phantom{.}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{......}
\end{array}\]
