Advertisements
Advertisements
Question
Solution
(i) Saturated Hydrocarbons: The compounds of carbon having only single bonds between the carbon atoms are called saturated compounds. This includes alkanes, having a general formula CnH2n+2.
(ii) Unsaturated Hydrocarbons: The compounds of carbon having double and triple bonds between the carbon atoms are called unsaturated compounds. This includes alkenes and alkynes having general formula CnH2n and CnH2n-2, respectively.
Example
(i) Saturated hydrocarbon: C2H6 (Ethane)
Structure:
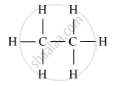
Structure:
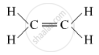
Structure:
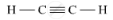
RELATED QUESTIONS
Explain in brief two main reasons for carbon forming a large number of compounds.
Draw the electron dot structures for ethanoic acid.
Draw the electron dot structures for H2S.
Fill in the following blank with suitable word:
The form of carbon which is used as a lubricant at high temperature is .........
Name a chemical reaction which is characteristic of unsaturated hydrocarbons (like alkenes and alkynes).
Explain the following reaction with balanced chemical equation:
Calcium reacts with water.
Which among the following are unsaturated hydrocarbons?
- \[\ce{H3C - CH2 - CH2 - CH3}\]
- \[\ce{H3C - C ≡ C - CH3}\]
- \[\begin{array}{cc}
\ce{H3C - CH - CH3}\\
|\phantom{..}\\
\phantom{.}\ce{CH3}
\end{array}\] - \[\begin{array}{cc}
\ce{H3C - C = CH2}\\
|\phantom{.}\\
\phantom{..}\ce{CH3}
\end{array}\]
Name the reaction which is commonly used in the conversion of vegetable oils to fats. Explain the reaction involved in detail.
Write the name of the following compound
\[\begin{array}{cc}
\phantom{}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{}\\
\ce{H - C - C - C - C - C - C - C = O}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{....}|\phantom{.....}\\
\phantom{.}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{......}
\end{array}\]
- Write the name and draw the structure of a saturated hydrocarbon with four carbon atoms.
- Write the number of single covalent bonds present in this compound.
