Advertisements
Advertisements
Question
Explain the nature of the covalent bond using the bond formation in CH3Cl.
Solution
The structure of CH3Cl is given below:
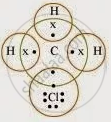
The molecule of chloromethane (CH₃Cl) is made up of three elements: carbon (Z = 6), hydrogen (Z = 1), and chlorine (Z = 17). Carbon has four valence electrons (2, 4), hydrogen has one (1), and chlorine has seven valence electrons (2, 8, 7). To achieve a full octet, carbon shares three of its valence electrons with three hydrogen atoms, and the fourth electron is shared with the chlorine atom.
APPEARS IN
RELATED QUESTIONS
What are covalent compounds?
Explain why carbon forms compounds mainly by covalent bond.
Give reason why carbon compounds are generally poor conductors of electricity.
What would be the electron dot structure of a molecule of sulphur which is made up of eight atoms of sulphur? (Hint – the eight atoms of sulphur are joined together in the form of a ring.)
State the type of bonding in the following molecule.
Water
What type of bonds are present in hydrogen chloride and oxygen?
What type of bonds are present in H2 molecule? Draw their electron-dot structures.
What type of bonds are present in Cl2 molecule? Draw their electron-dot structures.
Give one example of a molecule containing a double covalent bond
Explain the formation of a chlorine molecule on the basis of electronic theory of valency.
Give one example, state what are covalent compounds?
What is buckminsterfullerene?
Draw the electron-dot structure of HCl compound and state the type of bonding.
Draw the electron-dot structure of N2 and state the type of bonding.
Explain why, diamond can be used in rock drilling equipment but graphite cannot.
Why is graphite a good conductor of electricity but diamond is a non-conductor of electricity?
Buckminsterfullerene is an allotropic form of the element:
(a) phoshorus
(b) fluorine
(c) carbon
(d) sulphur
One of the following compounds is not ionic in nature. This compound is:
(a) Lithium chloride
(b) Ammonium chloride
(c) Calcium chloride
(d) Carbon tetrachloride
The molecules having triple bond in them are:
(a) oxygen and ethyne
(b) carbon dioxide and ammonia
(c) methane and ethene
(d) nitrogen and ethyne
The electronic configurations of two elements A and B are given below:
| A | 2, 6 |
| B | 2, 8, 1 |
(a) What type of chemical bond is formed between the two atoms of A?
(b) What type of chemical bond will be formed between the atoms of A and B?
What happens when methane (natural gas) burns in air? Write the chemical equation of the reaction involved.
Define a covalent bond.
Give two example in following case:
Liquid non polar compounds
Explain the following term with example.
Unsaturated hydrocarbon
The following structural formula belongs to which carbon compound?

Give examples for the following:
Two gaseous polar compounds.
Fill in the blank with correct word from the bracket.
Melting and boiling points of covalent compounds are generally ______ (low, high).
Give reason as to why hydrogen chloride can be termed as a polar covalent compound.
An element L consists of molecules.
What type of bonding is present in the particles that make up L?
The following table shows the electronic configuration of the elements W, X, Y, Z:
|
Element |
W |
X |
Y |
Z |
|
Electronic |
2,8,1 |
2,8,7 |
2,5 |
1 |
Answer the following questions based on the table above:
What type of bond is formed between Y and Z.
Which of the following is a common characteristic of a covalent compound?
Name greenish-yellow gas which also bleaches.
Generally, the melting and boiling point of carbon compounds are found to be less than _______ °C.
The bond between two oxygen atoms is _______ bond.
From the following hydrocarbon _______ is the cyclic hydrocarbon.
Write an Explanation.
Alkyne
Which of the following compound(s) possesses a high melting point?
Assertion (A): Melting point and boiling point of ethanol are lower than that of sodium chloride.
Reason (R): The forces of attraction between the molecules of ionic compounds are very strong.
Carbon can neither form C4- cation nor C4 anion. Why?
