Advertisements
Advertisements
Question
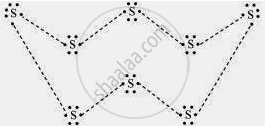
What would be the electron dot structure of a molecule of sulphur which is made up of eight atoms of sulphur? (Hint – the eight atoms of sulphur are joined together in the form of a ring.)
Solution
APPEARS IN
RELATED QUESTIONS
List three characteristic properties of covalent compounds.
What would be the electron dot structure of carbon dioxide which has the formula CO2?
Name the following:
The property of element by virtue of which atoms of the element can link to each other in the form of a long chain or ring structure.
Name the element whose one of the allotropic forms is buckminsterfullerene.
Buckminsterfullerene is spherical molecule in which 60 carbon atoms are arranged in interlocking hexagonal and pentagonal rings of carbon atoms.
How many hexagons of carbon atoms are present in one molecule of buckminsterfullerene?
What type of bonds are present in methane (CH4) and sodium chloride (NaCl)?
Fill in the following blank with suitable word:
The general formula CnH2n for cycloalkanes is the same as that of ...........
What type of chemical bonds are formed by carbon? Why?
Compare the properties of ionic compounds and covalent compounds.
What is buckminsterfullerene?
Why is diamond used for making cutting tools (like glass cutters) but graphite is not?
Using electron-dot diagrams which show only the outermost shell electrons, show how a molecule of nitrogen, N2, is formed from two nitrogen atoms. What name is given to this type of bonding? (Atomic number of nitrogen is 7)
Draw the electron-dot structure of NH3 and state the type of bonding.
State any two uses of graphite.
The number of covalent bonds in pentane (molecular formula C5H12) is:
The molecules having triple bond in them are:
(a) oxygen and ethyne
(b) carbon dioxide and ammonia
(c) methane and ethene
(d) nitrogen and ethyne
One of the following contains a double bond as well as single bonds. This is:
(a) CO2
(b) O2
(c) C2H4
(d) C2H2
An element A has 4 valence electrons in its atom whereas element B has only one valence electron in its atom. The compound formed by A and B does not conduct electricity. What is the nature of chemical bond in the compound formed? Give its electron-dot structure.
A solid element X has four electrons in the outermost shell of its atom. An allotrope Y of this element is used as a dry lubricant in machinery and also in making pencil leads.
(a) What is element X?
(b) Name the allotrope Y.
(c) State whether allotrope Y is a good conductor or non-conductor of electricity.
(d) Name one use of allotrope Y (other than lubrication and pencil leads)
(e) Name two other allotropes of element X.
What is the difference between ionic compounds and polar covalent compounds?
State the type of bonding in the following molecule.
Ammonium ion
Give two example in following case:
Liquid non polar compounds
Draw all possible structural formulae of compound from their molecular formula given below.
C3H8
Explain the structure of Hydronium ion.
Acids dissolve in water to produce positively charged ions. Draw the structure of these positive ions.
Explain why Carbon tetrachloride does not dissolve in water.
Name a neutral covalent molecule which contains one lone pair of electrons.
What is the term defined below?
A bond formed by a shared pair of electrons, each bonding atom contributing one electron to the pair.
Define the functional group and complete the following table.
| S.I. No. | Functional Group | Compound | Formula |
| (1) | ______________ | _______________ |
C2H5OH |
| (2) | _______________ | _______________ | CH3CHO |
Answer the following question.
What is methane? Draw its electron dot structure. Name the type of bonds formed in this compound. Why are such compounds:
(i) poor conductors of electricity? and
(ii) have low melting and boiling points? What happens when this compound burns in oxygen?
Write scientific reason.
Benzene compounds are called aromatic compounds.
Write a short note.
Aromatic hydrocarbons
Complete the following activity.
Write the names of the hydrocarbons for the following structural formula.
(isobutylene, cyclohexane, propene, cyclohexene, cyclopentane, benzene, propyne, isobutane, propane)
 |
Acetic acid was added to a solid X kept in a test tube. A colourless, odourless gas Y was evolved. The gas was passed through lime water, which turned milky. It was concluded that ______.
A molecule of ammonia (NH3) has
The table shows the electronic structures of four elements.
| Element | Electronic Structure |
| P | 2, 6 |
| Q | 2, 8, 1 |
| R | 2, 8, 7 |
| S | 2, 8, 8 |
- Identify which element(s) will form covalent bonds with carbon.
- “Carbon reacts with an element in the above table to form several compounds.” Give suitable reason.
______ is an example of a covalent compound having a high melting point.
In covalent bond formation, the sharing of ______ electrons takes place in their outermost shell.
Name the following:
\[\ce{CH3 - CH2CH = CH2}\]
