Advertisements
Advertisements
प्रश्न
What would be the electron dot structure of a molecule of sulphur which is made up of eight atoms of sulphur? (Hint – the eight atoms of sulphur are joined together in the form of a ring.)
उत्तर
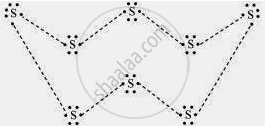
APPEARS IN
संबंधित प्रश्न
List three characteristic properties of covalent compounds.
Buckminsterfullerene is spherical molecule in which 60 carbon atoms are arranged in interlocking hexagonal and pentagonal rings of carbon atoms.
How many hexagons of carbon atoms are present in one molecule of buckminsterfullerene?
Buckminsterfullerene is spherical molecule in which 60 carbon atoms are arranged in interlocking hexagonal and pentagonal rings of carbon atoms.
How many pentagons of carbon atoms are present in one molecule of buckminsterfullerene?
What type of chemical bond is formed between carbon and bromine?
Give one example of a molecule containing a single covalent bond.
Give one example of a molecule containing a double covalent bond
Fill in the blank in the following sentence:
In forming N2 molecule, .............. electrons are shared by each atom of nitrogen.
What type of chemical bonds are formed by carbon? Why?
How will you find out which of the water soluble compound A or B is ionic?
Give the formula of the compound that would be formed by the combination of the following pair of elements:
K and H
Which inert gas does the Cl atom in HCl resemble in electron arrangement?
Explain why, graphite can be used as a lubricant but diamond cannot.
What is diamond? Of what substance is diamond made?
Why is graphite a good conductor of electricity but diamond is a non-conductor of electricity?
The number of covalent bonds in pentane (molecular formula C5H12) is:
A saturated hydrocarbon has fifty hydrogen atom in its molecule. The number of carbon atoms in its molecule will be
(a) twenty five
(b) twenty four
(c) twenty six
(d) twenty seven
The number of isomers formed by the hydrocarbon with molecular formula C5H12 is:
(a) 2
(b) 5
(c) 3
(d) 4
What is the difference between ionic compounds and polar covalent compounds?
State the type of bonding in the following molecule.
Hydroxyl ion
Complete the following:
In case of non-polar covalent bond, the covalent bond is formed in the ______ of atoms and shared electrons are ______ distributed. (corner, middle, equally, unequally)
Explain the following term with example.
Alkane
Explain the following term with example.
Homopolymer
Give examples for the following:
Two liquid non polar compounds.
Explain the following:
Polar covalent compounds are good conductors of electricity.
Name a neutral covalent molecule which contains one lone pair of electrons.
Compare the compounds carbon tetrachloride and sodium chloride with regard to solubility in water and electrical conductivity.
Draw an electron dot diagram to show the formation of the following compound.
Methane
Draw an electron dot diagram to show the formation of the following compound.
Magnesium chloride [ H=1, C=6, Mg=12, Cl=17].
Compound X consists of molecules.
Choose the letter corresponding to the correct answer from the options A, B, C and D given below:
X is likely to have a ______.
What is the difference between a polar covalent compound and a non-polar covalent compound?
Potassium (Atomic No. 19) and chlorine (Atomic No. 17) react to form a compound. On the basis of electronic concept, explain
(i) oxidation
(ii) reduction
(iii)oxidising agent
(iv)reducing agent
Name two compounds that are covalent when taken pure but produce ions when dissolved in water.
Define a covalent bond.
Name the types of Hydrocarbons.
Name two carbon compounds used in day-to-day life.
Considering MgCl2 as ionic compound and CH4 as covalent compound give any two differences between these two compounds.
Which of the following are correct structural isomers of butane?
- \[\begin{array}{cc}
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\
|\phantom{....}|\phantom{....}|\phantom{....}|\\
\ce{H - C - C - C - C - H}\\
|\phantom{....}|\phantom{....}|\phantom{....}|\\
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\
\end{array}\] - \[\begin{array}{cc}
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\
|\phantom{....}|\phantom{....}|\\
\ce{H - C - C - C - H}\\
|\phantom{.....}|\phantom{.....}|\\
\ce{H}\ce{H-C-H}\ce{H}\\
|\\
\ce{H}\\
\end{array}\] - \[\begin{array}{cc}
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\
|\phantom{....}|\phantom{....}|\\
\ce{H - C - C - C - H}\\
|\phantom{.....}\backslash\phantom{..}|\\
\phantom{....}\ce{H}\phantom{......}\ce{C - H}\phantom{}\\
\phantom{.......}|\\
\phantom{.......}\ce{H}\\
\end{array}\] - \[\begin{array}{cc}
\ce{H}\phantom{...}\ce{H}\\
|\phantom{....}|\\
\ce{H - C - C - H}\\
|\phantom{....}|\\
\ce{H - C - C - H}\\
|\phantom{....}|\\
\ce{H}\phantom{...}\ce{H}\\
\end{array}\]
"Carbon prefers to share its valence electrons with other atoms of carbon or with atoms of other elements rather than gaining or losing the valence electrons in order to attain noble gas configuration." Give reasons to justify this statement.
Carbon can neither form C4- cation nor C4 anion. Why?
