Advertisements
Advertisements
प्रश्न
Give one example of a molecule containing a double covalent bond
उत्तर
A molecule containing a double covalent bond: oxygen molecule 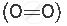
APPEARS IN
संबंधित प्रश्न
State the reason to explain why covalent compounds "are bad conductors of electricity".
Name a carbon containing molecule which has two double bonds.
What type of bonds are present in methane (CH4) and sodium chloride (NaCl)?
What type of chemical bonds are formed by carbon? Why?
What type of chemical bond is present in chlorine molecule? Explain your answer.
will CCl4 conduct electricity or not?
give reason for your choice.
Draw the electron-dot structure of HCl compound and state the type of bonding.
Explain why, diamond has a high melting point.
The atomic numbers of four elements P, Q, R and S are 6, 10, 12 and 17 respectively. Which two elements can combine to form a covalent compound?
(a) P and R
(b) Q and S
(c) P and S
(d) R and S
One of the following is not an allotrope of carbon. This is:
(a) diamond
(b) graphite
(c) cumene
(d) buckministerfullerene
Give two example in following case:
Gaseous polar compounds
(a) Compound X consists of molecules.
Choose the letter corresponding to the correct answer from the choices (a), (b), (c) and (d) given below
The type of bonding in X will be
An element L consists of molecules.
What type of bonding is present in the particles that make up L?
Draw the electron dot diagram and structure of :
methane
Write scientific reason.
Carbon has the property of forming many compounds.
Complete the following activity.
Write the names of the hydrocarbons for the following structural formula.
(isobutylene, cyclohexane, propene, cyclohexene, cyclopentane, benzene, propyne, isobutane, propane)
| \[\begin{array}{cc}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\ |\phantom{....}|\phantom{....}|\\\ce{H - C - C - C - H}\\ |\phantom{....}|\phantom{....}|\\\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H} \end{array}\] |
When ethyl alcohol and acetic acid are mixed, the resulting ester has a chemical formula ______.
Carbon forms four covalent bonds by sharing its four valence electrons with four univalent atoms, e.g. hydrogen. After the formation of four bonds, carbon attains the electronic configuration of
In covalent bond formation, the sharing of ______ electrons takes place in their outermost shell.
