Advertisements
Advertisements
प्रश्न
Give one example of a molecule containing a triple covalent bond.
उत्तर
A molecule containing a triple covalent bond: nitrogen molecule 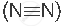 )
)
APPEARS IN
संबंधित प्रश्न
List three characteristic properties of covalent compounds.
What is buckminsterfullerene?
Explain why, diamond can be used in rock drilling equipment but graphite cannot.
Why is graphite a good conductor of electricity but diamond is a non-conductor of electricity?
The organic compound prepared by Wohler from an inorganic compound called ammonium cyanate was:
(a) glucose
(b) urea
(c) uric acid
(d) vinegar
The electronic configurations of two elements A and B are given below:
| A | 2, 6 |
| B | 2, 8, 1 |
(a) What type of chemical bond is formed between the two atoms of A?
(b) What type of chemical bond will be formed between the atoms of A and B?
Give the characteristic properties of covalent compounds.
Explain the following:
polar covalent compounds electricity.
For each compound mentioned above give the formulae of ions formed in aqueous solution.
Give two example in following case:
Liquid non polar compounds
Explain the following term with example.
Unsaturated hydrocarbon
State the type of bonding in the following molecule.
Water
Potassium chloride is an electrovalent compound, while hydrogen chloride is a covalent compound, But, both conduct electricity in their aqueous solutions. Explain.
Discuss in brief about the properties of coordinate covalent compounds.
Mineral acids are stronger acids than carboxylic acids because
- mineral acids are completely ionised
- carboxylic acids are completely ionised
- mineral acids are partially ionised
- carboxylic acids are partially ionised
Statement (A): Covalent compounds are bad conductors of electricity.
Reason (B): Covalent compounds contain charged particles (ions)
State the reasons, why carbon cannot
- Lose four electrons to form C4+ cation and
- Gain four electrons to form C4- anion.
How does carbon overcome this problem to form compounds?
It is observed that covalent compounds are had conductors of electricity. Give reason.
