Advertisements
Advertisements
Question
Give one example of a molecule containing a triple covalent bond.
Solution
A molecule containing a triple covalent bond: nitrogen molecule 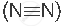 )
)
APPEARS IN
RELATED QUESTIONS
What do you call the compounds having the same molecular formula but different structural arrangements of atoms?
What type of bond exist in CCl4?
How will you find out which of the water soluble compound A or B is ionic?
Give the formula of the compound that would be formed by the combination of the following pair of elements:
Al and Cl2
Give the formula of the compound that would be formed by the combination of the following pair of elements:
K and H
Draw the electron-dot structure of NH3 and state the type of bonding.
A saturated hydrocarbon has fifty hydrogen atom in its molecule. The number of carbon atoms in its molecule will be
(a) twenty five
(b) twenty four
(c) twenty six
(d) twenty seven
The number of carbon atoms joined in a spherical molecule of buckminsterfullerene is:
(a) fifty
(b) sixty
(c) seventy
(d) ninety
What are the conditions necessary for the formation of covalent molecules?
Give the characteristic properties of covalent compounds.
Explain the following:
polar covalent compounds electricity.
Define a coordinate bond and give the conditions for its formation.
Draw an electron dot structure of the following molecule. (Without showing the circle) :
Ethene
Choose and write the correct option.
Which type of carbon-carbon bonds are present in Vanaspati ghee?
Explain the following briefly:
Cl2 is a non polar molecule, while HCl is a polar molecule.
What are the term defined below?
A bond formed by share pair of electrons, each bonding atom contributing one electron to the pair.
State the type of bond formed when the combining atom has zero E.N. difference.
Generally, the melting and boiling point of carbon compounds are found to be less than _______ °C.
Write scientific reason.
Carbon has the property of forming many compounds.
Which of the following is the formula of Butanoic acid?
