Advertisements
Advertisements
प्रश्न
Give one example of a molecule containing a triple covalent bond.
उत्तर
A molecule containing a triple covalent bond: nitrogen molecule 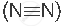 )
)
APPEARS IN
संबंधित प्रश्न
State the reason to explain why covalent compounds "are bad conductors of electricity".
Name the following:
The property of element by virtue of which atoms of the element can link to each other in the form of a long chain or ring structure.
Why are most carbon compounds poor conductors of electricity?
What is a covalent bond?
Friedrich Wohler converted an inorganic compound into an organic compound in the laboratory.
Write the name and formula of organic compound formed.
What type of bonding would you expect between Carbon and Chlorine?
Draw the electron-dot structure of NH3 and state the type of bonding.
Explain why, diamond has a high melting point.
Describe the structure of graphite with the help of a labelled diagram.
Match the pairs.
| Group 'A' | Group 'B' |
| a. C2H6 | 1. Unsaturated hydrocarbon |
| b. C2H2 | 2. Molecular formula of an alcohol |
| c. CH4O | 3. Saturated hydrocarbon |
| d. C3H6 | 4. Triple bond |
Draw an electron dot structure of the following molecule. (Without showing the circle) :
Methane
Draw an electron dot structure of the following molecule. (Without showing the circle) :
Ethene
Explain the Structural isomerism term with example.
What do you understand by lone pair and shared pair?
Fill in the blank with correct word from the bracket.
Melting and boiling points of covalent compounds are generally ______ (low, high).
State the type of bond formed when the combining atom has small E.N. difference.
Name the types of Hydrocarbons.
Which of the following is the formula of Butanoic acid?
"Carbon prefers to share its valence electrons with other atoms of carbon or with atoms of other elements rather than gaining or losing the valence electrons in order to attain noble gas configuration." Give reasons to justify this statement.
