Advertisements
Advertisements
Question
Give one example of a molecule containing a double covalent bond
Solution
A molecule containing a double covalent bond: oxygen molecule 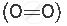
APPEARS IN
RELATED QUESTIONS
What would be the electron dot structure of carbon dioxide which has the formula CO2?
Butanone is a four-carbon compound with the functional group:
Choose the correct answer from the options given below:
Which of the following is a common characteristic of a covalent compound?
1) high melting point
2) consists of molecules
3) always soluble in water
4) Conducts electricity when it is in the molten state
An element L consists of molecules.
What type of bonding is present in the particles that make up L?
Give one example of a molecule containing a single covalent bond.
What type of chemical bond is present in chlorine molecule? Explain your answer.
Compare the properties of ionic compounds and covalent compounds.
State one test by which sodium chloride can be distinguished from sugar.
Explain why, diamond is hard while graphite is soft (though both are made of carbon atoms).
One of the following contains a double bond as well as single bonds. This is:
(a) CO2
(b) O2
(c) C2H4
(d) C2H2
Two non-metals combine with each other by the sharing of electrons to form a compound X.
(a) What type of chemical bond is present in X?
(b) State whether X will have a high melting point or low melting point.
(c) Will it be a good conductor of electricity or not?
(d) Will it dissolve in an organic solvent or not?
The electronic configurations of three elements X, Y and Z are as follows:
| X | 2, 4 |
| Y | 2, 7 |
| Z | 2, 1 |
(a) Which two elements will combine to form an ionic compound?
(b) Which two elements will react to form a covalent compound?
Give reasons for your choice.
What is the difference between ionic compounds and covalent compounds?
For each compound mentioned above give the formulae of ions formed in aqueous solution.
Define a coordinate bond and give the conditions for its formation.
Explain the following:
Covalent compounds have low melting and boiling point.
State the type of bond formed when the combining atom has small E.N. difference.
State the type of bond formed, and draw Lewis structure of water.
Write the molecular formula of the given compound.
Propylene
Write scientific reason.
Benzene compounds are called aromatic compounds.
