Advertisements
Advertisements
प्रश्न
Draw the electron-dot structure of HCl compound and state the type of bonding.
उत्तर
The electron-dot structure of HCl is:
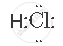
The hydrogen atom shares its one electron with a chlorine atom to form a covalent bond.
APPEARS IN
संबंधित प्रश्न
Name the type of bonds formed in ionic compounds and in the compounds formed by carbon.
State one major difference between covalent and ionic bonds and give one example each of covalent and ionic compounds.
What is the atomic number of carbon. Write its electronic configuration.
Give the formula of the compound that would be formed by the combination of the following pair of elements:
Al and Cl2
Draw the electron-dot structure of H2O compound and state the type of bonding.
Using electron-dot diagrams which show only the outermost shell electrons, show how a molecule of oxygen, O2, is formed from two oxygen atoms. What name is given to this type of bonding? (At. No. of oxygen = 8)
Explain why, diamond is hard while graphite is soft (though both are made of carbon atoms).
Draw the electron-dot structure of NH3 and state the type of bonding.
Explain why, graphite can be used as a lubricant but diamond cannot.
Explain why, diamond can be used in rock drilling equipment but graphite cannot.
Explain why, diamond has a high melting point.
what substance is graphite made?
The molecules having triple bond in them are:
(a) oxygen and ethyne
(b) carbon dioxide and ammonia
(c) methane and ethene
(d) nitrogen and ethyne
Explain the following term with example.
Unsaturated hydrocarbon
(a) Compound X consists of molecules.
Choose the letter corresponding to the correct answer from the choices (a), (b), (c) and (d) given below
X is likely to have a :
Compare the compounds carbon tetrachloride and sodium chloride with regard to solubility in water and electrical conductivity.
What is the difference between :
Ionic compounds and covalent compounds
Name greenish-yellow gas which also bleaches.
Molecular reactions are ______ in the covalent compound.
"Carbon prefers to share its valence electrons with other atoms of carbon or with atoms of other elements rather than gaining or losing the valence electrons in order to attain noble gas configuration." Give reasons to justify this statement.
