Advertisements
Advertisements
Question
Draw the electron-dot structure of HCl compound and state the type of bonding.
Solution
The electron-dot structure of HCl is:
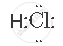
The hydrogen atom shares its one electron with a chlorine atom to form a covalent bond.
APPEARS IN
RELATED QUESTIONS
State whether the following statement is true or false:
Diamond and graphite are the covalent compounds of carbon element (C)
What type of bonds are present in Cl2 molecule? Draw their electron-dot structures.
You can buy solid air-freshners in shops. Do you think these substance are ionic or covalent? Why?
Give the formulae of the chlorides of the elements X and Y having atomic numbers of 3 and 6 respectively. Will the properties of the two chlorides be similar or different? Explain your answer.
State the type of bonding in the following molecule.
Water
Draw an electron dot diagram for the formation of the following. State the type of bonding present in them.
Hydroxyl ion
Compare the compounds carbon tetrachloride and sodium chloride with regard to solubility in water and electrical conductivity.
An element L consists of molecules.
What type of bonding is present in the particles that make up L?
Draw the electron dot structure of covalent compound methane (non polar) and HCL (polar) and give two difference between them.
Name greenish-yellow gas which also bleaches.
Name the anion present in the following compound:
When a barium chloride solution is added to a solution of compound B, a white precipitate insoluble in dilute hydrochloric acid is formed.
What are Allotropes? Name any two allotropic forms of carbon. Give one use of it.
The number of electrons in the valence shell of a carbon atom is 4.
Your body is made up of carbon.
Write an Explanation.
Alkane
Explain covalent bond with example.
An element A is soft and can be cut with a knife. This is very reactive to air and cannot be kept open in air. It reacts vigorously with water. Identify the element from the following
In covalent bond formation, the sharing of ______ electrons takes place in their outermost shell.
Non-polar covalent compounds are ______ conductors of heat and electricity.
