Advertisements
Advertisements
Question
Draw the electron-dot structure of H2O compound and state the type of bonding.
Solution
The electron-dot structure of H2O is:
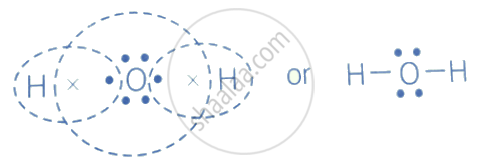
The oxygen atom shares its one electron with each hydrogen atom to form a covalent bond in the water molecule.
APPEARS IN
RELATED QUESTIONS
Why covalent compounds are different from ionic compounds?
Elements forming ionic compounds attain noble gas configuration by either gaining or losing electrons from their outermost shells. Give reason to explain why carbon cannot attain noble gas configuration in this manner to form its compounds.
Explain why, diamond can be used in rock drilling equipment but graphite cannot.
The solution of one of the following compounds will not conduct electricity. This compounds is:
(a) NaCl
(b) CCl4
(c) MgCl2
(d) CaCl2
One of the following compounds is not ionic in nature. This compound is:
(a) Lithium chloride
(b) Ammonium chloride
(c) Calcium chloride
(d) Carbon tetrachloride
The organic compound prepared by Wohler from an inorganic compound called ammonium cyanate was:
(a) glucose
(b) urea
(c) uric acid
(d) vinegar
What do you understand by polar covalent compounds?
Name the types of Hydrocarbons.
The number of electrons in the valence shell of a carbon atom is 4.
Write a short note.
Characteristics of carbon
Considering MgCl2 as ionic compound and CH4 as covalent compound give any two differences between these two compounds.
Give an example for each of the following statement
A compound in which two Covalent bonds are formed.
Discuss in brief about the properties of coordinate covalent compounds.
Cation is formed when ______.
Oils on treating with hydrogen in the presence of palladium or nickel catalyst form fats. This is an example of
Mineral acids are stronger acids than carboxylic acids because
- mineral acids are completely ionised
- carboxylic acids are completely ionised
- mineral acids are partially ionised
- carboxylic acids are partially ionised
Statement (A): Covalent compounds are bad conductors of electricity.
Reason (B): Covalent compounds contain charged particles (ions)
Non-polar covalent compounds are ______ conductors of heat and electricity.
In covalent compounds, the bond is formed due to ______ of electrons.
