Advertisements
Advertisements
प्रश्न
Draw the electron-dot structure of H2O compound and state the type of bonding.
उत्तर
The electron-dot structure of H2O is:
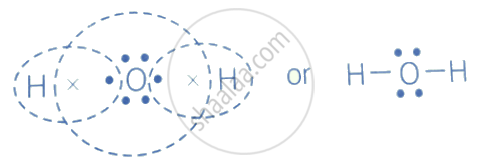
The oxygen atom shares its one electron with each hydrogen atom to form a covalent bond in the water molecule.
APPEARS IN
संबंधित प्रश्न
Write structural formula of Methane.
Elements forming ionic compounds attain noble gas configuration by either gaining or losing electrons from their outermost shells. Give reason to explain why carbon cannot attain noble gas configuration in this manner to form its compounds.
What type of bonds are present in methane (CH4) and sodium chloride (NaCl)?
Why is diamond used for making cutting tools (like glass cutters) but graphite is not?
Friedrich Wohler converted an inorganic compound into an organic compound in the laboratory.
Write the name and formula of organic compound formed.
will CCl4 conduct electricity or not?
give reason for your choice.
will CS2 conduct electricity or not?
give reason for your choice.
Why is graphite a good conductor of electricity but diamond is a non-conductor of electricity?
The electronic configurations of two elements A and B are given below:
| A | 2, 6 |
| B | 2, 8, 1 |
(a) What type of chemical bond is formed between the two atoms of A?
(b) What type of chemical bond will be formed between the atoms of A and B?
What do you understand by polar covalent compounds?
Give two example in following case:
Gaseous polar compounds
The covalent bond in which the electrons are shared equally between the combining atoms.
Compare the compounds carbon tetrachloride and sodium chloride with regard to solubility in water and electrical conductivity.
State the type of bond formed, and draw Lewis structure of water.
Explain covalent bond with example.
Write a short note.
Characteristics of carbon
Write a short note.
Aromatic hydrocarbons
Identify the incorrect statement and correct them.
- Like covalent compounds, coordinate compounds also contain charged particles (ions). So they are good conductors of electricity.
- Ionic bond is a weak bond when compared to Hydrogen bond.
- Ionic or electrovalent bonds are formed by mutual sharing of electrons between atoms.
- Loss of electrons is called Oxidation and gain of electron is called Reduction.
- The electrons which are not involved in bonding are called valence electrons.
Which of the following statements are usually correct for carbon compounds? These
- are good conductors of electricity
- are poor conductors of electricity
- have strong forces of attraction between their molecules
- do not have strong forces of attraction between their molecules.
Write notes on the characteristics of covalent compounds.
