Advertisements
Advertisements
प्रश्न
Draw the electron-dot structure of H2O compound and state the type of bonding.
उत्तर
The electron-dot structure of H2O is:
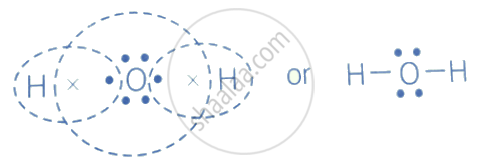
The oxygen atom shares its one electron with each hydrogen atom to form a covalent bond in the water molecule.
APPEARS IN
संबंधित प्रश्न
Write the electron-dot structure for the following molecule.
Cl2
Give one example of a molecule containing a triple covalent bond.
Fill in the blank in the following sentence:
Two atoms of the same element combine to form a molecule. The bond between them is known as ................ bond.
Why is graphite a good conductor of electricity but diamond is a non-conductor of electricity?
Out of KCl, HCl, CCl4 and NaCl, the compounds which are not ionic are:
(a) KCl and HCl
(b) HCl and CCl4
(c) CCl4 and NaCl
(d) KCl and CCl4
A hydrocarbon having one double bond has 100 carbon atoms in its molecule. The number of hydrogen atoms in its molecule will be
(a) 200
(b) 198
(c) 202
(d) 196
The number of carbon atoms joined in a spherical molecule of buckminsterfullerene is:
(a) fifty
(b) sixty
(c) seventy
(d) ninety
Define a covalent bond.
Give the characteristic properties of covalent compounds.
Draw all possible structural formulae of compound from their molecular formula given below.
C3H8
Observe the straight chain hydrocarbons given below and answer the following
questions :
i) Which of the straight chain compounds from A and B is saturated and unsaturated straight chains?
ii) Name these straight chains.
iii) Write their chemical formulae and number of – CH2 - units.

Explain the following briefly.
Pure water does not conduct electricity, but on adding sodium chloride to it, it starts conducting electricity.
Choose the correct answer from the options given below
Which one is not example of polar covalent compound?
Fill in the blank with correct word from the bracket.
Melting and boiling points of covalent compounds are generally ______ (low, high).
The particles present in strong electrolytes are
Electrons are getting added to an element Y:
Is Y getting oxidised or reduced?
Acids dissolve in water and produce positively charged ions. Draw the structure of these positive ions.
What is the difference between :
Ionic compounds and covalent compounds
The bond between two oxygen atoms is _______ bond.
Write the molecular formula of the given compound.
Propylene
