Advertisements
Advertisements
प्रश्न
Write the electron-dot structure for the following molecule.
Cl2
Atomic number of chlorine is 17.
Draw the electron dot structure of a chlorine molecule.
उत्तर
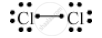
APPEARS IN
संबंधित प्रश्न
State the reason why carbon can neither form \[\ce{C^4+}\] cations nor \[\ce{C^4−}\] anions but forms covalent compound.
Name the black substance of pencil. Will the current flow through the electrical circuit when we use the sharpened ends of the pencil to complete the circuit?
What do you call the compounds having the same molecular formula but different structural arrangements of atoms?
Fill in the following blank with suitable word:
The form of carbon which is known as black lead is ...........
What type of bonding would you expect between Carbon and Chlorine?
Draw the electron-dot structure of N2 and state the type of bonding.
Write two points of difference in the structures of diamond and graphite.
What is diamond? Of what substance is diamond made?
The molecules having triple bond in them are:
(a) oxygen and ethyne
(b) carbon dioxide and ammonia
(c) methane and ethene
(d) nitrogen and ethyne
Give the characteristic properties of covalent compounds.
Define a coordinate bond and give the conditions for its formation.
Draw an electron dot diagram for the formation of the following. State the type of bonding present in them.
Hydroxyl ion
Explain the following term with example.
Alkane
Draw an electron dot diagram to show the formation of the following compound.
Magnesium chloride [ H=1, C=6, Mg=12, Cl=17].
What are the term defined below?
A bond formed by share pair of electrons, each bonding atom contributing one electron to the pair.
Element A has 2 electrons in its M shell. Element B has atomic number 7.
Write equations to show how A and B form ions.
Explain the following:
Polar covalent compounds conduct electricity?
Covalent bonds can be single, double or triple covalent bonds. How many electrons are shared in each? Give an example of each type.
Consider the coordination compound, K2[Cu(CN)4]. A coordinate covalent bond exists between:
Which of the following is the purest form of carbon?
