Advertisements
Advertisements
प्रश्न
Write the electron-dot structure for the following molecule.
Cl2
Atomic number of chlorine is 17.
Draw the electron dot structure of a chlorine molecule.
उत्तर
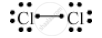
APPEARS IN
संबंधित प्रश्न
Why covalent compounds are different from ionic compounds?
State the reason to explain why covalent compounds "have low melting and boiling points."
Give appropriate scientific reasons for Carbon tetrachloride does not conduct electricity.
What type of bonds are present in water molecule? Draw the electron-dot structure of water (H2O).
Name one covalent compound containing chlorine.
The number of covalent bonds in pentane (molecular formula C5H12) is:
What are the conditions necessary for the formation of covalent molecules?
Give two example in following case:
Gaseous non polar compounds
Give examples for the following:
Two gaseous polar compounds.
The following table shows the electronic configuration of the elements W, X, Y, Z:
|
Element |
W |
X |
Y |
Z |
|
Electronic |
2,8,1 |
2,8,7 |
2,5 |
1 |
Answer the following questions based on the table above:
What type of bond is formed between Y and Z.
Which of the following is a common characteristic of a covalent compound?
Explain why carbon tetrachloride does not dissolve in water.
What is the difference between a polar covalent compound and a non-polar covalent compound?
In the formation of electrovalent compounds, electrons are transferred from one element to another. How are electrons involved in the formation of a covalent compound?
Answer the following question.
What is methane? Draw its electron dot structure. Name the type of bonds formed in this compound. Why are such compounds:
(i) poor conductors of electricity? and
(ii) have low melting and boiling points? What happens when this compound burns in oxygen?
Write an Explanation.
Alkene
Considering MgCl2 as ionic compound and CH4 as covalent compound give any two differences between these two compounds.
Which of the following statements are usually correct for carbon compounds? These
- are good conductors of electricity
- are poor conductors of electricity
- have strong forces of attraction between their molecules
- do not have strong forces of attraction between their molecules.
Two carbon atoms can always form one or two covalent bonds.
The table shows the electronic structures of four elements.
| Element | Electronic Structure |
| P | 2, 6 |
| Q | 2, 8, 1 |
| R | 2, 8, 7 |
| S | 2, 8, 8 |
- Identify which element(s) will form covalent bonds with carbon.
- “Carbon reacts with an element in the above table to form several compounds.” Give suitable reason.
