Advertisements
Advertisements
Questions
Write the electron-dot structure for the following molecule.
Cl2
Atomic number of chlorine is 17.
Draw the electron dot structure of a chlorine molecule.
Solution
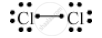
APPEARS IN
RELATED QUESTIONS
What would be the electron dot structure of carbon dioxide which has the formula CO2?
Choose the most appropriate answer from the following list of oxides which fit the description.
A covalent oxide of a metalloid.
State whether the following statement is true or false:
Diamond and graphite are the covalent compounds of carbon element (C)
Buckminsterfullerene is spherical molecule in which 60 carbon atoms are arranged in interlocking hexagonal and pentagonal rings of carbon atoms.
How many hexagons of carbon atoms are present in one molecule of buckminsterfullerene?
Give one example of a molecule containing a double covalent bond
Draw the electron-dot structure of H2O compound and state the type of bonding.
Explain why, graphite can be used as a lubricant but diamond cannot.
what substance is graphite made?
One of the following is not an allotrope of carbon. This is:
(a) diamond
(b) graphite
(c) cumene
(d) buckministerfullerene
Which of the following cannot exhibit isomerism?
(a) C4H10
(b) C5H12
(c) C3H8
(d) C6H14
An element E exists in three allotropic forms A, B and C. In allotrope A, the atoms of element E are joined to form spherical molecules. In allotrope B, each atom of element E is surrounded by three other E atoms to form a sheet like structure. In allotrope C, each atom of element E is surrounded by four other E atoms to form a rigid structure.
(a) Name the element E.
(b) What is allotrope A.
(c) What is allotrope B?
(d) What is allotrope C?
(e) Which allotrope is used in making jewellery?
(f) Which allotrope is used in making anode of a dry cell?
Explain the following term with example.
Unsaturated hydrocarbon
The carbon compound is used in daily life is ______.
Explain the following briefly.
Pure water does not conduct electricity, but on adding sodium chloride to it, it starts conducting electricity.
What do you understand by lone pair and shared pair?
Which of the following is a common characteristic of a covalent compound?
Potassium chloride is an electrovalent compound, while hydrogen chloride is a covalent compound, But, both conduct electricity in their aqueous solutions. Explain.
Write the molecular formula of the given compound.
Propylene
Two carbon atoms can always form one or two covalent bonds.
