Advertisements
Advertisements
Question
What type of bonds are present in water molecule? Draw the electron-dot structure of water (H2O).
Solution
The chemical bond present in a water molecule is covalent bond because one oxygen atom shares its two electrons with two hydrogen atoms.
The electron-dot structure of a water molecule is represented as:
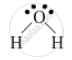
APPEARS IN
RELATED QUESTIONS
An element L consists of molecules.
What type of bonding is present in the particles that make up L?
Fill in the blank in the following sentence:
Two chlorine atoms combine to form a molecule. The bond between them is known as ..........
Friedrich Wohler converted an inorganic compound into an organic compound in the laboratory.
Write the name and formula of organic compound formed.
Draw the electron-dot structure of HCl compound and state the type of bonding.
what substance is graphite made?
Describe the structure of graphite with the help of a labelled diagram.
Why is graphite a good conductor of electricity but diamond is a non-conductor of electricity?
What are the conditions necessary for the formation of covalent molecules?
Methane molecule is non-polar molecule. Explain.
Draw an electron dot structure of the following molecule. (Without showing the circle) :
Methanol
Taking hydrogen chloride and methane as examples, distinguish between a polar covalent bond and a non polar covalent bond.
Explain the following:
Covalent compounds are generally gases or liquids or soft solids.
(a) Compound X consists of molecules.
Choose the letter corresponding to the correct answer from the choices (a), (b), (c) and (d) given below
In the liquid state, X will
Acids dissolve in water to produce positively charged ions. Draw the structure of these positive ions.
Explain the bonding in methane molecule using electron dot structure.
Which of the following is a common characteristic of a covalent compound?
In the formation of electrovalent compounds, electrons are transferred from one element to another. How are electrons involved in the formation of a covalent compound?
Complete the following activity.
Write the names of the hydrocarbons for the following structural formula.
(isobutylene, cyclohexane, propene, cyclohexene, cyclopentane, benzene, propyne, isobutane, propane)
 |
Although metals form basic oxides, which of the following metals form an amphoteric oxide?
Carbon forms four covalent bonds by sharing its four valence electrons with four univalent atoms, e.g. hydrogen. After the formation of four bonds, carbon attains the electronic configuration of
