Advertisements
Advertisements
प्रश्न
What type of bonds are present in water molecule? Draw the electron-dot structure of water (H2O).
उत्तर
The chemical bond present in a water molecule is covalent bond because one oxygen atom shares its two electrons with two hydrogen atoms.
The electron-dot structure of a water molecule is represented as:
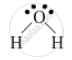
APPEARS IN
संबंधित प्रश्न
Ethane, with the molecular formula C2H6 has ______.
State the type of bonding in the following molecule.
Water
State whether the following statement is true or false:
Diamond and graphite are the covalent compounds of carbon element (C)
Buckminsterfullerene is spherical molecule in which 60 carbon atoms are arranged in interlocking hexagonal and pentagonal rings of carbon atoms.
How many hexagons of carbon atoms are present in one molecule of buckminsterfullerene?
State one major difference between covalent and ionic bonds and give one example each of covalent and ionic compounds.
What type of bond is present in oxygen molecule?
What type of chemical bond is present in chlorine molecule? Explain your answer.
What is buckminsterfullerene?
How buckminsterfullerene is it related to diamond and graphite?
Explain why, diamond is hard while graphite is soft (though both are made of carbon atoms).
State one use of diamond which depends on its 'extraordinary brilliance' and one use of graphite which depends on its being 'black and quite soft'.
Out of KCl, HCl, CCl4 and NaCl, the compounds which are not ionic are:
(a) KCl and HCl
(b) HCl and CCl4
(c) CCl4 and NaCl
(d) KCl and CCl4
The number of isomers formed by the hydrocarbon with molecular formula C5H12 is:
(a) 2
(b) 5
(c) 3
(d) 4
Draw an electron dot diagram for the formation of the following. State the type of bonding present in them.
Hydroxyl ion
Give two example in following case:
Solid covalent compounds
Explain the following:
Polar covalent compounds are good conductors of electricity.
Explain the bonding in methane molecule using electron dot structure.
State the type of bond formed when the combining atom has zero E.N. difference.
Although metals form basic oxides, which of the following metals form an amphoteric oxide?
Carbon can neither form C4- cation nor C4 anion. Why?
