Advertisements
Advertisements
प्रश्न
Explain the following:
Polar covalent compounds conduct electricity?
उत्तर
Polar covalent compounds conduct electricity because they form ions in their solutions and hence dissociate in water.
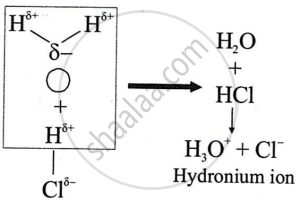
APPEARS IN
संबंधित प्रश्न
What type of bonds are present in CO2 molecule? Draw their electron-dot structures.
Buckminsterfullerene is an allotropic form of the element:
(a) phoshorus
(b) fluorine
(c) carbon
(d) sulphur
A solid element X has four electrons in the outermost shell of its atom. An allotrope Y of this element is used as a dry lubricant in machinery and also in making pencil leads.
(a) What is element X?
(b) Name the allotrope Y.
(c) State whether allotrope Y is a good conductor or non-conductor of electricity.
(d) Name one use of allotrope Y (other than lubrication and pencil leads)
(e) Name two other allotropes of element X.
Give two example in following case:
Liquid non polar compounds
Choose and write the correct option.
Which type of carbon-carbon bonds are present in Vanaspati ghee?
Give examples for the following:
Two liquid non polar compounds.
(a) Compound X consists of molecules.
Choose the letter corresponding to the correct answer from the choices (a), (b), (c) and (d) given below
In the liquid state, X will
Name a neutral covalent molecule which contains one lone pair of electrons.
Compound X consists of molecules.
Choose the letter corresponding to the correct answer from the options A, B, C and D given below:
X is likely to have a ______.
Which of the following are correct structural isomers of butane?
- \[\begin{array}{cc}
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\
|\phantom{....}|\phantom{....}|\phantom{....}|\\
\ce{H - C - C - C - C - H}\\
|\phantom{....}|\phantom{....}|\phantom{....}|\\
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\
\end{array}\] - \[\begin{array}{cc}
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\
|\phantom{....}|\phantom{....}|\\
\ce{H - C - C - C - H}\\
|\phantom{.....}|\phantom{.....}|\\
\ce{H}\ce{H-C-H}\ce{H}\\
|\\
\ce{H}\\
\end{array}\] - \[\begin{array}{cc}
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\
|\phantom{....}|\phantom{....}|\\
\ce{H - C - C - C - H}\\
|\phantom{.....}\backslash\phantom{..}|\\
\phantom{....}\ce{H}\phantom{......}\ce{C - H}\phantom{}\\
\phantom{.......}|\\
\phantom{.......}\ce{H}\\
\end{array}\] - \[\begin{array}{cc}
\ce{H}\phantom{...}\ce{H}\\
|\phantom{....}|\\
\ce{H - C - C - H}\\
|\phantom{....}|\\
\ce{H - C - C - H}\\
|\phantom{....}|\\
\ce{H}\phantom{...}\ce{H}\\
\end{array}\]
