Advertisements
Advertisements
Question
Explain the following:
Polar covalent compounds conduct electricity?
Solution
Polar covalent compounds conduct electricity because they form ions in their solutions and hence dissociate in water.
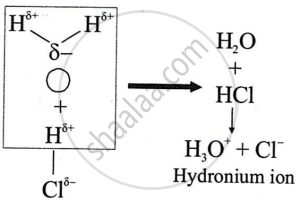
APPEARS IN
RELATED QUESTIONS
List three characteristic properties of covalent compounds.
State the reason why carbon can neither form \[\ce{C^4+}\] cations nor \[\ce{C^4−}\] anions but forms covalent compound.
Draw the electron-dot structure of a hydrogen chloride molecule.
Which inert gas does the H atom in HCl resemble in electron arrangement?
Which inert gas does the Cl atom in HCl resemble in electron arrangement?
The atomic numbers of four elements P, Q, R and S are 6, 10, 12 and 17 respectively. Which two elements can combine to form a covalent compound?
(a) P and R
(b) Q and S
(c) P and S
(d) R and S
The solution of one of the following compounds will not conduct electricity. This compounds is:
(a) NaCl
(b) CCl4
(c) MgCl2
(d) CaCl2
Explain the following briefly:
Sodium chloride dissolves in water but carbon tetra chloride is insoluble in water.
In the formation of electrovalent compounds, electrons are transferred from one element to another. How are electrons involved in the formation of a covalent compound?
Write an Explanation.
Alkyne
