Advertisements
Advertisements
Question
Draw the electron-dot structure of a hydrogen chloride molecule.
Solution
The electron-dot structure of a hydrogen chloride molecule is:
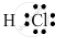
It is formed by sharing one electron by the hydrogen and chlorine atoms.
APPEARS IN
RELATED QUESTIONS
Elements forming ionic compounds attain noble gas configuration by either gaining or losing electrons from their outermost shells. Give reason to explain why carbon cannot attain noble gas configuration in this manner to form its compounds.
Explain the nature of the covalent bond using the bond formation in CH3Cl.
Name the following:
The property of element by virtue of which atoms of the element can link to each other in the form of a long chain or ring structure.
What type of chemical bond is formed between carbon and bromine?
What type of bonds are present in CH4 molecule? Draw their electron-dot structures.
Fill in the following blank with suitable word:
The form of carbon which is known as black lead is ...........
Compare the properties of ionic compounds and covalent compounds.
Name one covalent compound containing chlorine.
Draw the electron-dot structure of CO2 compound and state the type of bonding.
what substance is graphite made?
The number of covalent bonds in pentane (molecular formula C5H12) is:
The pencil leads are made of mainly:
(a) lithium
(b) charcoal
(c) lead
(d) graphite
State the type of bonding in the following molecule.
Hydroxyl ion
Explain the following term with example.
Homopolymer
Acids dissolve in water and produce positively charged ions. Draw the structure of these positive ions.
Name two compounds that are covalent when taken pure but produce ions when dissolved in water.
From the following hydrocarbon _______ is the cyclic hydrocarbon.
Which of the following compounds of carbon does not consist of ions?
Two carbon atoms can always form one or two covalent bonds.
Assertion (A): Melting point and boiling point of ethanol are lower than that of sodium chloride.
Reason (R): The forces of attraction between the molecules of ionic compounds are very strong.
