Advertisements
Advertisements
प्रश्न
Draw the electron-dot structure of a hydrogen chloride molecule.
उत्तर
The electron-dot structure of a hydrogen chloride molecule is:
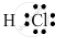
It is formed by sharing one electron by the hydrogen and chlorine atoms.
APPEARS IN
संबंधित प्रश्न
What are covalent compounds?
List three characteristic properties of covalent compounds.
State the reason to explain why covalent compounds "are bad conductors of electricity".
What type of bonds are present in water molecule? Draw the electron-dot structure of water (H2O).
What type of bonds are present in methane (CH4) and sodium chloride (NaCl)?
Fill in the following blank with suitable word:
The form of carbon which is known as black lead is ...........
Which inert gas electron configuration do the Cl atoms in Cl2 molecule resemble? What is this electron configuration?
Explain the formation of a chlorine molecule on the basis of electronic theory of valency.
Name one covalent compound containing chlorine.
Explain why, diamond is hard while graphite is soft (though both are made of carbon atoms).
Explain the following term with example.
Alkane
Observe the straight chain hydrocarbons given below and answer the following
questions :
i) Which of the straight chain compounds from A and B is saturated and unsaturated straight chains?
ii) Name these straight chains.
iii) Write their chemical formulae and number of – CH2 - units.

Explain the following briefly.
Pure water does not conduct electricity, but on adding sodium chloride to it, it starts conducting electricity.
An element L consists of molecules.
Why L is heated with iron metal, it forms a compound FeL. What chemical term would you use to describe the change undergone by L?
Elements Q and S react together to form an ionic compound. Under normal conditions, which physical state will the compound QS exist in?
Number of valence electrons in a carbon atom is _______.
Your body is made up of carbon.
Complete the following activity.
Write the names of the hydrocarbons for the following structural formula.
(isobutylene, cyclohexane, propene, cyclohexene, cyclopentane, benzene, propyne, isobutane, propane)
 |
Give an example for each of the following statement.
Formation of coordinate covalent bond between NH3 ➝ BF3 molecules
