Advertisements
Advertisements
Question
What type of bonds are present in CH4 molecule? Draw their electron-dot structures.
Solution
Covalent bond is the chemical bond present in CH4 as a carbon atom shares its electrons with four hydrogen atoms.
The electron-dot structure is:
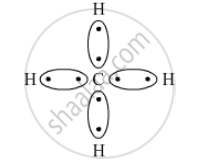
APPEARS IN
RELATED QUESTIONS
Give appropriate scientific reasons for Carbon tetrachloride does not conduct electricity.
How will you find out which of the water soluble compound A or B is ionic?
Draw the electron-dot structure of HCl compound and state the type of bonding.
Why is graphite a good conductor of electricity but diamond is a non-conductor of electricity?
The atomic numbers of four elements P, Q, R and S are 6, 10, 12 and 17 respectively. Which two elements can combine to form a covalent compound?
(a) P and R
(b) Q and S
(c) P and S
(d) R and S
One of the following compounds is not ionic in nature. This compound is:
(a) Lithium chloride
(b) Ammonium chloride
(c) Calcium chloride
(d) Carbon tetrachloride
A covalent molecule having a double bond between its atoms is:
(a) Hydrogen
(b) Oxygen
(c) water
(d) ammonia
The molecules having triple bond in them are:
(a) oxygen and ethyne
(b) carbon dioxide and ammonia
(c) methane and ethene
(d) nitrogen and ethyne
The electronic configurations of two elements A and B are given below:
| A | 2, 6 |
| B | 2, 8, 1 |
(a) What type of chemical bond is formed between the two atoms of A?
(b) What type of chemical bond will be formed between the atoms of A and B?
Distinguish between ionic and covalent compounds under the following properties:
(i) Strength of forces between constituent elements
(ii) Solubility of compounds in water
(iii) Electrical conduction in substances
Give examples for the following:
Two solid covalent compounds.
Give examples for the following:
Two gaseous non polar compounds.
Explain the following:
Polar covalent compounds are good conductors of electricity.
What do you understand by dipole (polar) molecule?
State the type of bond formed when the combining atom has small E.N. difference.
Write the molecular formula of the given compound.
Acetic acid
Complete the following activity.
Write the names of the hydrocarbons for the following structural formula.
(isobutylene, cyclohexane, propene, cyclohexene, cyclopentane, benzene, propyne, isobutane, propane)
| \[\begin{array}{cc}\phantom{......}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{..}\\ \phantom{.....}|\phantom{....}|\phantom{....}|\\ \ce{H - C - C = C}\\\phantom{.....}|\phantom{.........}|\\ \phantom{.....}\ce{H}\phantom{........}\ce{H}\end{array}\] |
Give an example for each of the following statement.
Formation of coordinate covalent bond between NH3 ➝ BF3 molecules
The number of pi-bonds present in benzoic acid molecule are ______.
The table shows the electronic structures of four elements.
| Element | Electronic Structure |
| P | 2, 6 |
| Q | 2, 8, 1 |
| R | 2, 8, 7 |
| S | 2, 8, 8 |
- Identify which element(s) will form covalent bonds with carbon.
- “Carbon reacts with an element in the above table to form several compounds.” Give suitable reason.
