Advertisements
Advertisements
Question
What type of bonds are present in Cl2 molecule? Draw their electron-dot structures.
Solution
Covalent bond is the chemical bond present in a Cl2 molecule as one chlorine atom shares its electrons with another chlorine atom.
The electron-dot structure is:
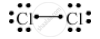
APPEARS IN
RELATED QUESTIONS
Name the hardest natural substance known.
What type of bonds are present in H2 molecule? Draw their electron-dot structures.
Fill in the blank in the following sentence:
In forming N2 molecule, .............. electrons are shared by each atom of nitrogen.
What is a covalent bond?
Why is graphite used for making dry cell electrodes but diamond is not?
The solution of one of the following compounds will not conduct electricity. This compounds is:
(a) NaCl
(b) CCl4
(c) MgCl2
(d) CaCl2
Out of KCl, HCl, CCl4 and NaCl, the compounds which are not ionic are:
(a) KCl and HCl
(b) HCl and CCl4
(c) CCl4 and NaCl
(d) KCl and CCl4
A covalent molecule having a double bond between its atoms is:
(a) Hydrogen
(b) Oxygen
(c) water
(d) ammonia
The number of isomers formed by the hydrocarbon with molecular formula C5H12 is:
(a) 2
(b) 5
(c) 3
(d) 4
What do you understand by polar covalent compounds?
For each compound mentioned above give the formulae of ions formed in aqueous solution.
Taking hydrogen chloride and methane as examples, distinguish between a polar covalent bond and a non polar covalent bond.
Explain the following:
Polar covalent compounds are good conductors of electricity.
Draw an electron dot diagram to show the formation of the following compound.
Methane
Define a covalent bond.
Give an example for each of the following statement.
A compound in which three covalent bonds are formed.
Identify the incorrect statement and correct them.
- Like covalent compounds, coordinate compounds also contain charged particles (ions). So they are good conductors of electricity.
- Ionic bond is a weak bond when compared to Hydrogen bond.
- Ionic or electrovalent bonds are formed by mutual sharing of electrons between atoms.
- Loss of electrons is called Oxidation and gain of electron is called Reduction.
- The electrons which are not involved in bonding are called valence electrons.
"Carbon prefers to share its valence electrons with other atoms of carbon or with atoms of other elements rather than gaining or losing the valence electrons in order to attain noble gas configuration." Give reasons to justify this statement.
The number of single and double bonds present in a molecule of benzene (C6H6) respectively, are ______.
