Advertisements
Advertisements
Question
What type of bonds are present in CO2 molecule? Draw their electron-dot structures.
Solution
Covalent bond is the bond present in O2 as one oxygen atom shares its electrons with another oxygen atom.
The electron-dot structure is:
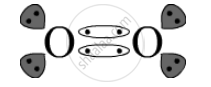
APPEARS IN
RELATED QUESTIONS
What are covalent compounds?
Explain the nature of the covalent bond using the bond formation in CH3Cl.
Name the black substance of pencil. Will the current flow through the electrical circuit when we use the sharpened ends of the pencil to complete the circuit?
What type of bonds are present in CH4 molecule? Draw their electron-dot structures.
Fill in the blank in the following sentence:
In forming N2 molecule, .............. electrons are shared by each atom of nitrogen.
Fill in the blank in the following sentence:
The number of single covalent bonds in C2H2 molecule are ...........
Why is graphite used for making dry cell electrodes but diamond is not?
Friedrich Wohler converted an inorganic compound into an organic compound in the laboratory.
Write the name and formula of organic compound formed.
Explain why, graphite can be used as a lubricant but diamond cannot.
Which of the following has a triple bond as well as single bonds?
(a) ethene
(b) methane
(c) ethyne
(d) nitrogen
What do you understand by polar covalent compounds?
Draw all possile structural formulae of compound from their molecular formula given below.
C3H4
What are the term defined below?
A bond formed by share pair of electrons, each bonding atom contributing one electron to the pair.
Name greenish-yellow gas which also bleaches.
Name the types of Hydrocarbons.
Your body is made up of carbon.
Give an example for each of the following statement.
A compound in which three covalent bonds are formed.
Which of the following is the purest form of carbon?
Which of the following is the formula of Butanoic acid?
In covalent compounds, the bond is formed due to ______ of electrons.
