Advertisements
Advertisements
Question
Which of the following has a triple bond as well as single bonds?
(a) ethene
(b) methane
(c) ethyne
(d) nitrogen
Solution
(c) ethyne
An ethyne molecule 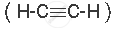 contains a triple bond between two carbon atoms and two single bonds between carbon and hydrogen atoms.
contains a triple bond between two carbon atoms and two single bonds between carbon and hydrogen atoms.
APPEARS IN
RELATED QUESTIONS
Fill in the following blank with suitable word:
The form of carbon which is known as black lead is ...........
What is the atomic number of carbon. Write its electronic configuration.
What type of bond is present in oxygen molecule?
What type of chemical bond is present in chlorine molecule? Explain your answer.
Friedrich Wohler converted an inorganic compound into an organic compound in the laboratory.
Write the name and formula of organic compound formed.
How will you find out which of the water soluble compound A or B is ionic?
Draw the electron-dot structure of CO2 compound and state the type of bonding.
Which of the following cannot exhibit isomerism?
(a) C4H10
(b) C5H12
(c) C3H8
(d) C6H14
The electronic configurations of two elements A and B are given below:
| A | 2, 6 |
| B | 2, 8, 1 |
(a) What type of chemical bond is formed between the two atoms of A?
(b) What type of chemical bond will be formed between the atoms of A and B?
You can buy solid air-freshners in shops. Do you think these substance are ionic or covalent? Why?
What are the conditions necessary for the formation of covalent molecules?
What is the difference between ionic compounds and covalent compounds?
Give reason as to why hydrogen chloride can be termed as a polar covalent compound.
State the type of bonding in the following molecule.
Water
Taking MgCl2 as an electrovalent compound, CCl4 as a covalent compound, give four difference between electrovalent and covalent compounds
Give an example for each of the following statement
A compound in which two Covalent bonds are formed.
Discuss in brief about the properties of coordinate covalent compounds.
Carbon forms four covalent bonds by sharing its four valence electrons with four univalent atoms, e.g. hydrogen. After the formation of four bonds, carbon attains the electronic configuration of
Carbon can neither form C4- cation nor C4 anion. Why?
