Advertisements
Advertisements
प्रश्न
Which of the following has a triple bond as well as single bonds?
(a) ethene
(b) methane
(c) ethyne
(d) nitrogen
उत्तर
(c) ethyne
An ethyne molecule 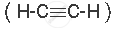 contains a triple bond between two carbon atoms and two single bonds between carbon and hydrogen atoms.
contains a triple bond between two carbon atoms and two single bonds between carbon and hydrogen atoms.
APPEARS IN
संबंधित प्रश्न
State whether the following statement is true or false:
Diamond and graphite are the covalent compounds of carbon element (C)
Give one example of a molecule containing a double covalent bond
Fill in the blanks in the following sentence:
In forming oxygen molecule, .............. electrons are shared by each atom of oxygen.
How buckminsterfullerene is it related to diamond and graphite?
Friedrich Wohler converted an inorganic compound into an organic compound in the laboratory.
Write the name and formula of organic compound formed.
Give the formulae of the chlorides of the elements X and Y having atomic numbers of 3 and 6 respectively. Will the properties of the two chlorides be similar or different? Explain your answer.
Complete the following:
When the nuclei of two different reacting atoms are of ______ mass, then a bond so formed is called ______ covalent bond. (equal, unequal, polar, non-polar)
Give two example in following case:
Gaseous polar compounds
The carbon compound is used in daily life is ______.
Explain the following briefly.
Pure water does not conduct electricity, but on adding sodium chloride to it, it starts conducting electricity.
Name a neutral covalent molecule which contains one lone pair of electrons.
State the type of bonding in the following molecule.
Water
Draw the electron dot structure of covalent compound methane (non polar) and HCL (polar) and give two difference between them.
Name gas with rotten egg smell.
Number of valence electrons in a carbon atom is _______.
Write a short note.
Aromatic hydrocarbons
Complete the following activity.
Write the names of the hydrocarbons for the following structural formula.
(isobutylene, cyclohexane, propene, cyclohexene, cyclopentane, benzene, propyne, isobutane, propane)
| \[\begin{array}{cc} \phantom{..}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\ \phantom{..}|\phantom{....}|\phantom{....}|\\ \ce{H - C - C- C- H}\\ \phantom{.}|\phantom{....}|\phantom{....}|\\ \ce{H - C - H}\\ |\\\ce{H}\end{array}\] |
Although metals form basic oxides, which of the following metals form an amphoteric oxide?
In covalent bond formation, the sharing of ______ electrons takes place in their outermost shell.
