Advertisements
Advertisements
प्रश्न
Which of the following has a triple bond as well as single bonds?
(a) ethene
(b) methane
(c) ethyne
(d) nitrogen
उत्तर
(c) ethyne
An ethyne molecule 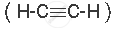 contains a triple bond between two carbon atoms and two single bonds between carbon and hydrogen atoms.
contains a triple bond between two carbon atoms and two single bonds between carbon and hydrogen atoms.
APPEARS IN
संबंधित प्रश्न
How buckminsterfullerene is it related to diamond and graphite?
Why is diamond used for making cutting tools (like glass cutters) but graphite is not?
will CCl4 conduct electricity or not?
give reason for your choice.
Using electron-dot diagrams which show only the outermost shell electrons, show how a molecule of nitrogen, N2, is formed from two nitrogen atoms. What name is given to this type of bonding? (Atomic number of nitrogen is 7)
Why are covalent compounds generally poor conductors of electricity?
A covalent molecule having a double bond between its atoms is:
(a) Hydrogen
(b) Oxygen
(c) water
(d) ammonia
The number of isomers formed by the hydrocarbon with molecular formula C5H12 is:
(a) 2
(b) 5
(c) 3
(d) 4
The electronic configurations of two elements A and B are given below:
| A | 2, 6 |
| B | 2, 8, 1 |
(a) What type of chemical bond is formed between the two atoms of A?
(b) What type of chemical bond will be formed between the atoms of A and B?
Match the pairs.
| Group 'A' | Group 'B' |
| a. C2H6 | 1. Unsaturated hydrocarbon |
| b. C2H2 | 2. Molecular formula of an alcohol |
| c. CH4O | 3. Saturated hydrocarbon |
| d. C3H6 | 4. Triple bond |
Draw all possile structural formulae of compound from their molecular formula given below.
C3H4
Explain the following briefly:
Cl2 is a non polar molecule, while HCl is a polar molecule.
Give examples for the following:
Two gaseous non polar compounds.
Explain the following:
Non-polar covalent compounds are insoluble in water.
Compare the compounds carbon tetrachloride and sodium chloride with regard to solubility in water and electrical conductivity.
Draw the electron dot diagram and structure of magnesium chloride.
What is the difference between :
Ionic compounds and covalent compounds
Answer the following question.
What is methane? Draw its electron dot structure. Name the type of bonds formed in this compound. Why are such compounds:
(i) poor conductors of electricity? and
(ii) have low melting and boiling points? What happens when this compound burns in oxygen?
Which of the following compounds of carbon does not consist of ions?
Statement (A): Covalent compounds are bad conductors of electricity.
Reason (B): Covalent compounds contain charged particles (ions)
Assertion (A): Melting point and boiling point of ethanol are lower than that of sodium chloride.
Reason (R): The forces of attraction between the molecules of ionic compounds are very strong.
