Advertisements
Advertisements
प्रश्न
What type of bonds are present in CO2 molecule? Draw their electron-dot structures.
उत्तर
Covalent bond is the bond present in O2 as one oxygen atom shares its electrons with another oxygen atom.
The electron-dot structure is:
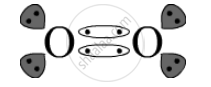
APPEARS IN
संबंधित प्रश्न
Write the number of covalent bonds in the molecule of butane, C4H10.
Name the type of bonds formed in ionic compounds and in the compounds formed by carbon.
How does graphite act as a lubricant?
What type of bonds are present in water molecule? Draw the electron-dot structure of water (H2O).
What type of chemical bond is present in chlorine molecule? Explain your answer.
How buckminsterfullerene is it related to diamond and graphite?
Which inert gas does the H atom in HCl resemble in electron arrangement?
Draw the electron-dot structure of H2O compound and state the type of bonding.
What is diamond? Of what substance is diamond made?
A covalent molecule having a double bond between its atoms is:
(a) Hydrogen
(b) Oxygen
(c) water
(d) ammonia
One of the following is not an allotrope of carbon. This is:
(a) diamond
(b) graphite
(c) cumene
(d) buckministerfullerene
State the type of bonding in the following molecule.
Ammonium ion
Match the pairs.
| Group 'A' | Group 'B' |
| a. C2H6 | 1. Unsaturated hydrocarbon |
| b. C2H2 | 2. Molecular formula of an alcohol |
| c. CH4O | 3. Saturated hydrocarbon |
| d. C3H6 | 4. Triple bond |
Choose the correct answer from the options given below
Condition favorable for formation of a covalent bond is
Taking MgCl2 as an electrovalent compound, CCl4 as a covalent compound, give four difference between electrovalent and covalent compounds
From the following hydrocarbon _______ is the cyclic hydrocarbon.
Consider the coordination compound, K2[Cu(CN)4]. A coordinate covalent bond exists between:
Which of the following compound(s) possesses a high melting point?
Molecular reactions are ______ in the covalent compound.
In covalent compounds, the bond is formed due to ______ of electrons.
