Advertisements
Advertisements
प्रश्न
What type of bonds are present in Cl2 molecule? Draw their electron-dot structures.
उत्तर
Covalent bond is the chemical bond present in a Cl2 molecule as one chlorine atom shares its electrons with another chlorine atom.
The electron-dot structure is:
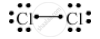
APPEARS IN
संबंधित प्रश्न
State the reason why carbon can neither form \[\ce{C^4+}\] cations nor \[\ce{C^4−}\] anions but forms covalent compound.
How does graphite act as a lubricant?
Name the hardest natural substance known.
What type of bonds are present in CO2 molecule? Draw their electron-dot structures.
Draw the electron-dot structure of a hydrogen chloride molecule.
Which inert gas does the H atom in HCl resemble in electron arrangement?
Draw the electron-dot structure of HCl compound and state the type of bonding.
Explain why, diamond can be used in rock drilling equipment but graphite cannot.
What is the difference between ionic compounds and polar covalent compounds?
Give reason as to why hydrogen chloride can be termed as a polar covalent compound.
Define a covalent bond.
Name greenish-yellow gas which also bleaches.
Name the anion present in the following compound:
When a barium chloride solution is added to a solution of compound B, a white precipitate insoluble in dilute hydrochloric acid is formed.
The bond between two oxygen atoms is _______ bond.
The molecular masses of a carbon compound spread over a range of _______.
Write an Explanation.
Alkane
Complete the following activity.
Write the names of the hydrocarbons for the following structural formula.
(isobutylene, cyclohexane, propene, cyclohexene, cyclopentane, benzene, propyne, isobutane, propane)
| \[\begin{array}{cc}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\ |\phantom{....}|\phantom{....}|\\\ce{H - C - C - C - H}\\ |\phantom{....}|\phantom{....}|\\\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H} \end{array}\] |
Complete the following activity.
Write the names of the hydrocarbons for the following structural formula.
(isobutylene, cyclohexane, propene, cyclohexene, cyclopentane, benzene, propyne, isobutane, propane)
| \[\begin{array}{cc} \phantom{.........}\ce{H}\\ \phantom{.........}|\\ \ce{H - C ≡ C - C - H}\\ \phantom{.........}|\\ \phantom{.........}\ce{H} \end{array}\] |
Complete the following activity.
Write the names of the hydrocarbons for the following structural formula.
(isobutylene, cyclohexane, propene, cyclohexene, cyclopentane, benzene, propyne, isobutane, propane)
 |
Which of the following is the formula of Butanoic acid?
