Advertisements
Advertisements
प्रश्न
Draw the electron-dot structure of HCl compound and state the type of bonding.
उत्तर
The electron-dot structure of HCl is:
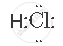
The hydrogen atom shares its one electron with a chlorine atom to form a covalent bond.
APPEARS IN
संबंधित प्रश्न
Elements forming ionic compounds attain noble gas configuration by either gaining or losing electrons from their outermost shells. Give reason to explain why carbon cannot attain noble gas configuration in this manner to form its compounds.
Name the black substance of pencil. Will the current flow through the electrical circuit when we use the sharpened ends of the pencil to complete the circuit?
How does graphite act as a lubricant?
Fill in the blank in the following sentence:
The number of single covalent bonds in C2H2 molecule are ...........
Draw the electron-dot structure of N2 and state the type of bonding.
State one use of diamond which depends on its 'extraordinary brilliance' and one use of graphite which depends on its being 'black and quite soft'.
What is graphite?
The solution of one of the following compounds will not conduct electricity. This compounds is:
(a) NaCl
(b) CCl4
(c) MgCl2
(d) CaCl2
Explain the Structural isomerism term with example.
Explain the following term with example.
Covalent bond
Explain the following:
Covalent compounds have low melting and boiling point.
Explain the following:
Polar covalent compounds are good conductors of electricity.
Choose the correct answer from the options given below
Condition favorable for formation of a covalent bond is
Draw the electron dot diagram and structure of :
methane
What is the difference between a polar covalent compound and a non-polar covalent compound?
What are Allotropes? Name any two allotropic forms of carbon. Give one use of it.
Define the functional group and complete the following table.
| S.I. No. | Functional Group | Compound | Formula |
| (1) | ______________ | _______________ |
C2H5OH |
| (2) | _______________ | _______________ | CH3CHO |
The bond between two oxygen atoms is _______ bond.
Write the molecular formula of the given compound.
Propylene
State the reasons, why carbon cannot
- Lose four electrons to form C4+ cation and
- Gain four electrons to form C4- anion.
How does carbon overcome this problem to form compounds?
