Advertisements
Advertisements
प्रश्न
What type of bonds are present in Cl2 molecule? Draw their electron-dot structures.
उत्तर
Covalent bond is the chemical bond present in a Cl2 molecule as one chlorine atom shares its electrons with another chlorine atom.
The electron-dot structure is:
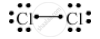
APPEARS IN
संबंधित प्रश्न
Write structural formula of Methane.
Why are most carbon compounds poor conductors of electricity?
State whether the following statement is true or false:
Diamond and graphite are the covalent compounds of carbon element (C)
Buckminsterfullerene is spherical molecule in which 60 carbon atoms are arranged in interlocking hexagonal and pentagonal rings of carbon atoms.
How many hexagons of carbon atoms are present in one molecule of buckminsterfullerene?
What type of bonds are present in methane (CH4) and sodium chloride (NaCl)?
What type of chemical bond is present in chlorine molecule? Explain your answer.
Describe the structure of diamond. Draw a simple diagram to show the arrangement of carbon atoms in diamond.
Buckminsterfullerene is an allotropic form of the element:
(a) phoshorus
(b) fluorine
(c) carbon
(d) sulphur
One of the following compounds is not ionic in nature. This compound is:
(a) Lithium chloride
(b) Ammonium chloride
(c) Calcium chloride
(d) Carbon tetrachloride
One of the following contains a double bond as well as single bonds. This is:
(a) CO2
(b) O2
(c) C2H4
(d) C2H2
You can buy solid air-freshners in shops. Do you think these substance are ionic or covalent? Why?
Explain the following term with example.
Homopolymer
The carbon compound is used in daily life is ______.
The following table shows the electronic configuration of the elements W, X, Y, Z:
|
Element |
W |
X |
Y |
Z |
|
Electronic |
2,8,1 |
2,8,7 |
2,5 |
1 |
Answer the following questions based on the table above:
What type of bond is formed between Y and Z.
Which of the following is a common characteristic of a covalent compound?
Elements Q and S react together to form an ionic compound. Under normal conditions, which physical state will the compound QS exist in?
Draw the electron dot diagram and structure of magnesium chloride.
Generally, the melting and boiling point of carbon compounds are found to be less than _______ °C.
The number of electrons in the valence shell of a carbon atom is 4.
Which of the following compound(s) possesses a high melting point?
