Advertisements
Advertisements
प्रश्न
What type of bonds are present in CH4 molecule? Draw their electron-dot structures.
उत्तर
Covalent bond is the chemical bond present in CH4 as a carbon atom shares its electrons with four hydrogen atoms.
The electron-dot structure is:
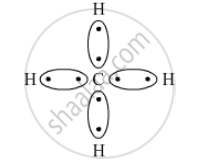
APPEARS IN
संबंधित प्रश्न
What is buckminsterfullerene?
The molecules having triple bond in them are:
(a) oxygen and ethyne
(b) carbon dioxide and ammonia
(c) methane and ethene
(d) nitrogen and ethyne
Give the formulae of the chlorides of the elements X and Y having atomic numbers of 3 and 6 respectively. Will the properties of the two chlorides be similar or different? Explain your answer.
What is the difference between ionic compounds and covalent compounds?
Give two example in following case:
Gaseous polar compounds
Explain the following term with example.
Hetero atom in a carbon compound
Explain the following briefly:
Sodium chloride dissolves in water but carbon tetra chloride is insoluble in water.
Explain why Carbon tetrachloride does not dissolve in water.
Name a neutral covalent molecule which contains one lone pair of electrons.
Draw an electron dot diagram to show the formation of the following compound.
Magnesium chloride [ H=1, C=6, Mg=12, Cl=17].
Element A has 2 electrons in its M shell. Element B has atomic number 7.
Write equations to show how A and B form ions.
Explain the following:
Polar covalent compounds conduct electricity?
Potassium chloride is an electrovalent compound, while hydrogen chloride is a covalent compound, But, both conduct electricity in their aqueous solutions. Explain.
In the formation of electrovalent compounds, electrons are transferred from one element to another. How are electrons involved in the formation of a covalent compound?
The number of electrons in the valence shell of a carbon atom is 4.
Write an Explanation.
Alkane
Which of the following compounds of carbon does not consist of ions?
The correct structural formula of butanoic acid is
The electron dot structure of chlorine molecule is:
Name the following:
\[\ce{CH3 - CH2CH = CH2}\]
