Advertisements
Advertisements
प्रश्न
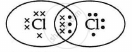
The electron dot structure of chlorine molecule is:
विकल्प
उत्तर
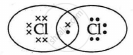
Explanation:
Cl2 is the molecule of chlorine. Chlorine has an electronic configuration of 2, 8, and 7. Here, a combination of two chlorine atoms with 7 valence electrons each occurs by sharing 1 electron.
APPEARS IN
संबंधित प्रश्न
List three characteristic properties of covalent compounds.
Why are most carbon compounds poor conductors of electricity?
State whether the following statement is true or false:
Diamond and graphite are the covalent compounds of carbon element (C)
What would be the electron-dot structure of carbon dioxide which has the formula CO2?
Fill in the following blank with suitable word:
The form of carbon which is known as black lead is ...........
Explain the formation of a chlorine molecule on the basis of electronic theory of valency.
Which inert gas does the H atom in HCl resemble in electron arrangement?
The pencil leads are made of mainly:
(a) lithium
(b) charcoal
(c) lead
(d) graphite
Define a covalent bond.
Draw all possible structural formulae of compound from their molecular formula given below.
C3H8
Give examples for the following:
Two solid covalent compounds.
The electronic configuration of N2 is 2, 5. How many electrons in the outer shell of a N atom are not involved in the formation of a nitrogen molecule?
State the type of bond formed when the combining atom has zero E.N. difference.
Define the functional group and complete the following table.
| S.I. No. | Functional Group | Compound | Formula |
| (1) | ______________ | _______________ |
C2H5OH |
| (2) | _______________ | _______________ | CH3CHO |
Although metals form basic oxides, which of the following metals form an amphoteric oxide?
______ is an example of a covalent compound having a high melting point.
Write notes on the characteristics of covalent compounds.