Advertisements
Advertisements
Question
The electron dot structure of chlorine molecule is:
Options
Solution
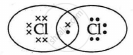
Explanation:
Cl2 is the molecule of chlorine. Chlorine has an electronic configuration of 2, 8, and 7. Here, a combination of two chlorine atoms with 7 valence electrons each occurs by sharing 1 electron.
APPEARS IN
RELATED QUESTIONS
Write structural formula of Methane.
Ethane, with the molecular formula C2H6 has ______.
What type of bonds are present in water molecule? Draw the electron-dot structure of water (H2O).
Fill in the following blank with suitable word:
The general formula CnH2n for cycloalkanes is the same as that of ...........
What is a covalent bond?
Draw the electron-dot structure of CO2 compound and state the type of bonding.
Explain the following:
polar covalent compounds electricity.
Give two example in following case:
Gaseous polar compounds
Draw all possible structural formulae of compound from their molecular formula given below.
C3H8
Give examples for the following:
Two solid covalent compounds.
What do you understand by lone pair and shared pair?
Explain the following:
Covalent compounds are generally gases or liquids or soft solids.
The particles present in strong electrolytes are
Define a covalent bond.
The bond between two oxygen atoms is _______ bond.
From the following hydrocarbon _______ is the cyclic hydrocarbon.
List two differences between the properties exhibited by covalent compounds and ionic compounds.
Which of the following is the formula of Butanoic acid?
A molecule of ammonia (NH3) has
"Carbon prefers to share its valence electrons with other atoms of carbon or with atoms of other elements rather than gaining or losing the valence electrons in order to attain noble gas configuration." Give reasons to justify this statement.