Advertisements
Advertisements
प्रश्न
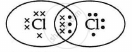
The electron dot structure of chlorine molecule is:
पर्याय
उत्तर
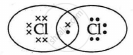
Explanation:
Cl2 is the molecule of chlorine. Chlorine has an electronic configuration of 2, 8, and 7. Here, a combination of two chlorine atoms with 7 valence electrons each occurs by sharing 1 electron.
APPEARS IN
संबंधित प्रश्न
Elements forming ionic compounds attain noble gas configuration by either gaining or losing electrons from their outermost shells. Give reason to explain why carbon cannot attain noble gas configuration in this manner to form its compounds.
Buckminsterfullerene is spherical molecule in which 60 carbon atoms are arranged in interlocking hexagonal and pentagonal rings of carbon atoms.
How many hexagons of carbon atoms are present in one molecule of buckminsterfullerene?
What do you call the compounds having the same molecular formula but different structural arrangements of atoms?
The number of covalent bonds in pentane (molecular formula C5H12) is:
Which of the following has a triple bond as well as single bonds?
(a) ethene
(b) methane
(c) ethyne
(d) nitrogen
One of the following is not an allotrope of carbon. This is:
(a) diamond
(b) graphite
(c) cumene
(d) buckministerfullerene
Give the characteristic properties of covalent compounds.
State the type of bonding in the following molecule.
Hydroxyl ion
Give two example in following case:
Gaseous polar compounds
Explain the following briefly:
Cl2 is a non polar molecule, while HCl is a polar molecule.
Acids dissolve in water to produce positively charged ions. Draw the structure of these positive ions.
Which of the following is a common characteristic of a covalent compound?
State the type of bond formed, and draw Lewis structure of water.
Fill in the blank and rewrite the completed statement:
Covalent compounds are generally soluble in _________ solvents.
Discuss in brief about the properties of coordinate covalent compounds.
______ is an example of a covalent compound having a high melting point.
Statement (A): Covalent compounds are bad conductors of electricity.
Reason (B): Covalent compounds contain charged particles (ions)
Name the following:
\[\ce{CH3 - CH2CH = CH2}\]
Non-polar covalent compounds are ______ conductors of heat and electricity.