Advertisements
Advertisements
Question
Draw an electron dot diagram for the formation of the following. State the type of bonding present in them.
Hydroxyl ion
Solution
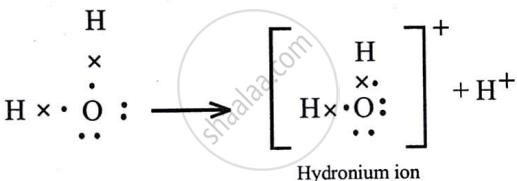
It has polar covalent bond.
APPEARS IN
RELATED QUESTIONS
Name the type of bonds formed in ionic compounds and in the compounds formed by carbon.
Ethane, with the molecular formula C2H6 has ______.
Buckminsterfullerene is spherical molecule in which 60 carbon atoms are arranged in interlocking hexagonal and pentagonal rings of carbon atoms.
How many pentagons of carbon atoms are present in one molecule of buckminsterfullerene?
What is the difference between ionic compounds and polar covalent compounds?
Explain the following term with example.
Homopolymer
Name a neutral covalent molecule which contains one lone pair of electrons.
Draw the electron dot structure of covalent compound methane (non polar) and HCL (polar) and give two difference between them.
Explain the following:
Polar covalent compounds conduct electricity?
The table shows the electronic structures of four elements.
| Element | Electronic Structure |
| P | 2, 6 |
| Q | 2, 8, 1 |
| R | 2, 8, 7 |
| S | 2, 8, 8 |
- Identify which element(s) will form covalent bonds with carbon.
- “Carbon reacts with an element in the above table to form several compounds.” Give suitable reason.
Molecular reactions are ______ in the covalent compound.
