Advertisements
Advertisements
प्रश्न
Draw an electron dot diagram for the formation of the following. State the type of bonding present in them.
Hydroxyl ion
उत्तर
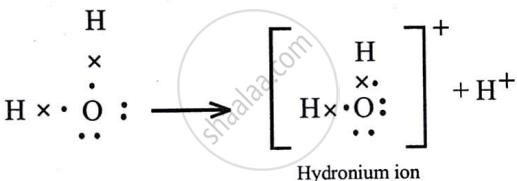
It has polar covalent bond.
APPEARS IN
संबंधित प्रश्न
An element L consists of molecules.
What type of bonding is present in the particles that make up L?
How does graphite act as a lubricant?
Write the electron-dot structure for the following molecule.
Cl2
Give the formula of the compound that would be formed by the combination of the following pair of elements:
Al and Cl2
The number of covalent bonds in pentane (molecular formula C5H12) is:
The number of carbon atoms joined in a spherical molecule of buckminsterfullerene is:
(a) fifty
(b) sixty
(c) seventy
(d) ninety
Complete the following:
When the nuclei of two different reacting atoms are of ______ mass, then a bond so formed is called ______ covalent bond. (equal, unequal, polar, non-polar)
Draw electron - dot structure and structural formula of methane.
The following table shows the electronic configuration of the elements W, X, Y, Z:
|
Element |
W |
X |
Y |
Z |
|
Electronic |
2,8,1 |
2,8,7 |
2,5 |
1 |
Answer the following questions based on the table above:
What type of bond is formed between Y and Z.
`"CH"_3 - "CH"_2 - "OH" overset("Alkaline""KMnO"_4 + "Heat")(->) "CH"_3 - "COOH"`
In the above given reaction, alkaline KMnO4 acts as
