Advertisements
Advertisements
प्रश्न
What would be the electron dot structure of a molecule of sulphur which is made up of eight atoms of sulphur? (Hint – the eight atoms of sulphur are joined together in the form of a ring.)
उत्तर
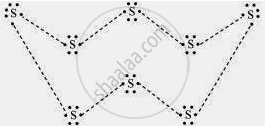
APPEARS IN
संबंधित प्रश्न
State the type of bonding in the following molecule.
Water
Choose the most appropriate answer from the following list of oxides which fit the description.
A covalent oxide of a metalloid.
How does graphite act as a lubricant?
What type of bonds are present in Cl2 molecule? Draw their electron-dot structures.
Fill in the blank in the following sentence:
In forming N2 molecule, .............. electrons are shared by each atom of nitrogen.
What is a covalent bond?
Give one example, state what are covalent compounds?
Why is diamond used for making cutting tools (like glass cutters) but graphite is not?
Why is graphite used for making dry cell electrodes but diamond is not?
State one test by which sodium chloride can be distinguished from sugar.
Give the formula of the compound that would be formed by the combination of the following pair of elements:
K and H
Which inert gas does the H atom in HCl resemble in electron arrangement?
Using electron-dot diagrams which show only the outermost shell electrons, show how a molecule of nitrogen, N2, is formed from two nitrogen atoms. What name is given to this type of bonding? (Atomic number of nitrogen is 7)
Draw the electron-dot structure of H2O compound and state the type of bonding.
What is graphite?
A saturated hydrocarbon has fifty hydrogen atom in its molecule. The number of carbon atoms in its molecule will be
(a) twenty five
(b) twenty four
(c) twenty six
(d) twenty seven
Which of the following cannot exhibit isomerism?
(a) C4H10
(b) C5H12
(c) C3H8
(d) C6H14
What is the difference between ionic compounds and polar covalent compounds?
Explain the following:
polar covalent compounds electricity.
Complete the following:
In case of non-polar covalent bond, the covalent bond is formed in the ______ of atoms and shared electrons are ______ distributed. (corner, middle, equally, unequally)
Give two example in following case:
Solid covalent compounds
Distinguish between ionic and covalent compounds under the following properties:
(i) Strength of forces between constituent elements
(ii) Solubility of compounds in water
(iii) Electrical conduction in substances
Draw an electron dot structure of the following molecule. (Without showing the circle) :
Ethene
Taking hydrogen chloride and methane as examples, distinguish between a polar covalent bond and a non polar covalent bond.
Explain the following briefly.
Pure water does not conduct electricity, but on adding sodium chloride to it, it starts conducting electricity.
What do you understand by lone pair and shared pair?
The molecule which contains a triple covalent bond is ______.
Covalent bonds can be single, double or triple covalent bonds. How many electrons are shared in each? Give an example of each type.
Name two compounds that are covalent when taken pure but produce ions when dissolved in water.
Define the functional group and complete the following table.
| S.I. No. | Functional Group | Compound | Formula |
| (1) | ______________ | _______________ |
C2H5OH |
| (2) | _______________ | _______________ | CH3CHO |
The molecular masses of a carbon compound spread over a range of _______.
Write scientific reason.
Benzene compounds are called aromatic compounds.
Complete the following activity.
Write the names of the hydrocarbons for the following structural formula.
(isobutylene, cyclohexane, propene, cyclohexene, cyclopentane, benzene, propyne, isobutane, propane)
| \[\begin{array}{cc} \phantom{.........}\ce{H}\\ \phantom{.........}|\\ \ce{H - C ≡ C - C - H}\\ \phantom{.........}|\\ \phantom{.........}\ce{H} \end{array}\] |
Complete the following activity.
Write the names of the hydrocarbons for the following structural formula.
(isobutylene, cyclohexane, propene, cyclohexene, cyclopentane, benzene, propyne, isobutane, propane)
 |
Complete the following activity.
Write the names of the hydrocarbons for the following structural formula.
(isobutylene, cyclohexane, propene, cyclohexene, cyclopentane, benzene, propyne, isobutane, propane)
 |
Give an example for each of the following statement.
Formation of coordinate covalent bond between NH3 ➝ BF3 molecules
"Carbon prefers to share its valence electrons with other atoms of carbon or with atoms of other elements rather than gaining or losing the valence electrons in order to attain noble gas configuration." Give reasons to justify this statement.
