Advertisements
Advertisements
प्रश्न
What would be the electron dot structure of carbon dioxide which has the formula CO2?
उत्तर
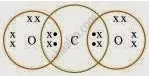
APPEARS IN
संबंधित प्रश्न
Elements forming ionic compounds attain noble gas configuration by either gaining or losing electrons from their outermost shells. Give reason to explain why carbon cannot attain noble gas configuration in this manner to form its compounds.
Choose the correct answer from the options given below:
Which of the following is a common characteristic of a covalent compound?
1) high melting point
2) consists of molecules
3) always soluble in water
4) Conducts electricity when it is in the molten state
What type of bonds are present in water molecule? Draw the electron-dot structure of water (H2O).
Fill in the blank in the following sentence:
The number of single covalent bonds in C2H2 molecule are ...........
What is the atomic number of carbon. Write its electronic configuration.
What type of bond exist in CCl4?
Give two general properties of ionic compounds and two those of covalent compounds.
will CCl4 conduct electricity or not?
give reason for your choice.
Name one covalent compound containing chlorine.
Which inert gas does the H atom in HCl resemble in electron arrangement?
What is diamond? Of what substance is diamond made?
State any two uses of graphite.
What are the conditions necessary for the formation of covalent molecules?
Explain the following:
polar covalent compounds electricity.
Draw an electron dot diagram for the formation of the following. State the type of bonding present in them.
Hydroxyl ion
Draw an electron dot structure of the following molecule. (Without showing the circle) :
Methane
Draw an electron dot structure of the following molecule. (Without showing the circle) :
Methanol
Explain the following term with example.
Covalent bond
Molecular formula of Propane is C3H8 , write the structural formula of propane.
Give examples for the following:
Two gaseous polar compounds.
Explain the structure of Ammonium ion.
Choose the correct answer from the options given below
Which one is not example of polar covalent compound?
(a) Compound X consists of molecules.
Choose the letter corresponding to the correct answer from the choices (a), (b), (c) and (d) given below
The type of bonding in X will be
An element L consists of molecules.
What type of bonding is present in the particles that make up L?
The particles present in strong electrolytes are
State the type of bonding in the following molecule.
Water
Draw the electron dot diagram and structure of nitrogen molecule.
Draw the electron dot diagram and structure of magnesium chloride.
What is the difference between :
Ionic compounds and covalent compounds
Covalent bonds can be single, double or triple covalent bonds. How many electrons are shared in each? Give an example of each type.
Write an Explanation.
Alkane
Complete the following activity.
Write the names of the hydrocarbons for the following structural formula.
(isobutylene, cyclohexane, propene, cyclohexene, cyclopentane, benzene, propyne, isobutane, propane)
| \[\begin{array}{cc} \phantom{..}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\ \phantom{..}|\phantom{....}|\phantom{....}|\\ \ce{H - C - C- C- H}\\ \phantom{.}|\phantom{....}|\phantom{....}|\\ \ce{H - C - H}\\ |\\\ce{H}\end{array}\] |
List two differences between the properties exhibited by covalent compounds and ionic compounds.
Cation is formed when ______.
It is observed that covalent compounds are had conductors of electricity. Give reason.
