Advertisements
Advertisements
प्रश्न
What would be the electron dot structure of carbon dioxide which has the formula CO2?
उत्तर
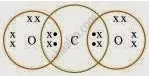
APPEARS IN
संबंधित प्रश्न
List three characteristic properties of covalent compounds.
Choose the most appropriate answer from the following list of oxides which fit the description.
A covalent oxide of a metalloid.
Name the following:
The property of element by virtue of which atoms of the element can link to each other in the form of a long chain or ring structure.
What type of bonds are present in H2 molecule? Draw their electron-dot structures.
Fill in the blanks in the following sentence:
In forming oxygen molecule, .............. electrons are shared by each atom of oxygen.
What type of bond exist in CCl4?
Friedrich Wohler converted an inorganic compound into an organic compound in the laboratory.
Write the name and formula of organic compound formed.
How will you find out which of the water soluble compound A or B is ionic?
Draw the electron-dot structure of a hydrogen chloride molecule.
What type of bonding would you expect between Carbon and Chlorine?
Draw the electron-dot structure of CO2 compound and state the type of bonding.
What is diamond? Of what substance is diamond made?
Explain why, diamond has a high melting point.
State any two uses of diamond.
What is graphite?
State any two uses of graphite.
Buckminsterfullerene is an allotropic form of the element:
(a) phoshorus
(b) fluorine
(c) carbon
(d) sulphur
Two non-metals combine with each other by the sharing of electrons to form a compound X.
(a) What type of chemical bond is present in X?
(b) State whether X will have a high melting point or low melting point.
(c) Will it be a good conductor of electricity or not?
(d) Will it dissolve in an organic solvent or not?
A solid element X has four electrons in the outermost shell of its atom. An allotrope Y of this element is used as a dry lubricant in machinery and also in making pencil leads.
(a) What is element X?
(b) Name the allotrope Y.
(c) State whether allotrope Y is a good conductor or non-conductor of electricity.
(d) Name one use of allotrope Y (other than lubrication and pencil leads)
(e) Name two other allotropes of element X.
An element E exists in three allotropic forms A, B and C. In allotrope A, the atoms of element E are joined to form spherical molecules. In allotrope B, each atom of element E is surrounded by three other E atoms to form a sheet like structure. In allotrope C, each atom of element E is surrounded by four other E atoms to form a rigid structure.
(a) Name the element E.
(b) What is allotrope A.
(c) What is allotrope B?
(d) What is allotrope C?
(e) Which allotrope is used in making jewellery?
(f) Which allotrope is used in making anode of a dry cell?
What do you understand by polar covalent compounds?
Give two example in following case:
Gaseous polar compounds
Explain the following term with example.
Homopolymer
Give examples for the following:
Two solid covalent compounds.
Explain the following:
Covalent compounds have low melting and boiling point.
Explain the following:
Non-polar covalent compounds are insoluble in water.
State the type of bond formed when the combining atom has small E.N. difference.
Draw the electron dot structure of covalent compound methane (non polar) and HCL (polar) and give two difference between them.
Covalent bonds can be single, double or triple covalent bonds. How many electrons are shared in each? Give an example of each type.
Taking MgCl2 as an electrovalent compound, CCl4 as a covalent compound, give four difference between electrovalent and covalent compounds
Name the anion present in the following compound:
When a barium chloride solution is added to a solution of compound B, a white precipitate insoluble in dilute hydrochloric acid is formed.
Name two carbon compounds used in day-to-day life.
Write the molecular formula of the given compound.
Propylene
Write a short note.
Aromatic hydrocarbons
Acetic acid was added to a solid X kept in a test tube. A colourless, odourless gas Y was evolved. The gas was passed through lime water, which turned milky. It was concluded that ______.
Which of the following is the formula of Butanoic acid?
`"CH"_3 - "CH"_2 - "OH" overset("Alkaline""KMnO"_4 + "Heat")(->) "CH"_3 - "COOH"`
In the above given reaction, alkaline KMnO4 acts as
The table shows the electronic structures of four elements.
| Element | Electronic Structure |
| P | 2, 6 |
| Q | 2, 8, 1 |
| R | 2, 8, 7 |
| S | 2, 8, 8 |
- Identify which element(s) will form covalent bonds with carbon.
- “Carbon reacts with an element in the above table to form several compounds.” Give suitable reason.
"Carbon prefers to share its valence electrons with other atoms of carbon or with atoms of other elements rather than gaining or losing the valence electrons in order to attain noble gas configuration." Give reasons to justify this statement.
