Advertisements
Advertisements
प्रश्न
Draw the electron-dot structure of CO2 compound and state the type of bonding.
उत्तर
The electron-dot structure of CO2 is:
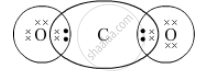
Both oxygen and carbon share their two electrons to form a covalent bond in carbon dioxide, CO2.
APPEARS IN
संबंधित प्रश्न
Name the type of bonds formed in ionic compounds and in the compounds formed by carbon.
Explain the nature of the covalent bond using the bond formation in CH3Cl.
Name the element whose one of the allotropic forms is buckminsterfullerene.
Why is graphite used for making dry cell electrodes but diamond is not?
What type of bonding would you expect between Carbon and Chlorine?
Using electron-dot diagrams which show only the outermost shell electrons, show how a molecule of oxygen, O2, is formed from two oxygen atoms. What name is given to this type of bonding? (At. No. of oxygen = 8)
The electronic configurations of three elements X, Y and Z are as follows:
| X | 2, 4 |
| Y | 2, 7 |
| Z | 2, 1 |
(a) Which two elements will combine to form an ionic compound?
(b) Which two elements will react to form a covalent compound?
Give reasons for your choice.
The electronic configurations of two elements A and B are given below:
| A | 2, 6 |
| B | 2, 8, 1 |
(a) What type of chemical bond is formed between the two atoms of A?
(b) What type of chemical bond will be formed between the atoms of A and B?
Draw an electron dot structure of the following molecule. (Without showing the circle) :
Water
Draw all possible structural formulae of compound from their molecular formula given below.
C3H8
Molecular formula of Propane is C3H8 , write the structural formula of propane.
Choose the correct answer from the options given below
Condition favorable for formation of a covalent bond is
The particles present in strong electrolytes are
Your body is made up of carbon.
Write an Explanation.
Alkane
Complete the following activity.
Write the names of the hydrocarbons for the following structural formula.
(isobutylene, cyclohexane, propene, cyclohexene, cyclopentane, benzene, propyne, isobutane, propane)
| \[\begin{array}{cc} \phantom{..}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\ \phantom{..}|\phantom{....}|\phantom{....}|\\ \ce{H - C - C- C- H}\\ \phantom{.}|\phantom{....}|\phantom{....}|\\ \ce{H - C - H}\\ |\\\ce{H}\end{array}\] |
The correct structural formula of butanoic acid is -
Which of the following compounds of carbon does not consist of ions?
In covalent bond formation, the sharing of ______ electrons takes place in their outermost shell.
Show the covalent bond formation in nitrogen molecule.
