Advertisements
Advertisements
प्रश्न
Explain the nature of the covalent bond using the bond formation in CH3Cl.
उत्तर
The structure of CH3Cl is given below:
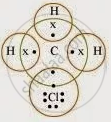
The molecule of chloromethane (CH₃Cl) is made up of three elements: carbon (Z = 6), hydrogen (Z = 1), and chlorine (Z = 17). Carbon has four valence electrons (2, 4), hydrogen has one (1), and chlorine has seven valence electrons (2, 8, 7). To achieve a full octet, carbon shares three of its valence electrons with three hydrogen atoms, and the fourth electron is shared with the chlorine atom.
APPEARS IN
संबंधित प्रश्न
Give reason why carbon compounds are generally poor conductors of electricity.
Choose the most appropriate answer from the following list of oxides which fit the description.
A covalent oxide of a metalloid.
Name the black substance of pencil. Will the current flow through the electrical circuit when we use the sharpened ends of the pencil to complete the circuit?
What do you call the compounds having the same molecular formula but different structural arrangements of atoms?
State one major difference between covalent and ionic bonds and give one example each of covalent and ionic compounds.
What type of bonds are present in H2 molecule? Draw their electron-dot structures.
Fill in the blank in the following sentence:
In forming N2 molecule, .............. electrons are shared by each atom of nitrogen.
What is a covalent bond?
will CS2 conduct electricity or not?
give reason for your choice.
Draw the electron-dot structure of a hydrogen chloride molecule.
Draw the electron-dot structure of HCl compound and state the type of bonding.
Draw the electron-dot structure of N2 and state the type of bonding.
Why is graphite a good conductor of electricity but diamond is a non-conductor of electricity?
The solution of one of the following compounds will not conduct electricity. This compounds is:
(a) NaCl
(b) CCl4
(c) MgCl2
(d) CaCl2
A saturated hydrocarbon has fifty hydrogen atom in its molecule. The number of carbon atoms in its molecule will be
(a) twenty five
(b) twenty four
(c) twenty six
(d) twenty seven
Which of the following cannot exhibit isomerism?
(a) C4H10
(b) C5H12
(c) C3H8
(d) C6H14
You can buy solid air-freshners in shops. Do you think these substance are ionic or covalent? Why?
What are the conditions necessary for the formation of covalent molecules?
What is the difference between ionic compounds and polar covalent compounds?
State the type of bonding in the following molecule.
Ammonium ion
Distinguish between ionic and covalent compounds under the following properties:
(i) Strength of forces between constituent elements
(ii) Solubility of compounds in water
(iii) Electrical conduction in substances
Explain the following term with example.
Covalent bond
Explain the following term with example.
Hetero atom in a carbon compound
Molecular formula of Propane is C3H8 , write the structural formula of propane.
(a) Compound X consists of molecules.
Choose the letter corresponding to the correct answer from the choices (a), (b), (c) and (d) given below
X is likely to have a :
An element L consists of molecules.
Why L is heated with iron metal, it forms a compound FeL. What chemical term would you use to describe the change undergone by L?
State the type of bond formed, and draw Lewis structure of water.
Name two compounds that are covalent when taken pure but produce ions when dissolved in water.
What are Allotropes? Name any two allotropic forms of carbon. Give one use of it.
The bond between two oxygen atoms is _______ bond.
The molecular masses of a carbon compound spread over a range of _______.
Complete the following activity.
Write the names of the hydrocarbons for the following structural formula.
(isobutylene, cyclohexane, propene, cyclohexene, cyclopentane, benzene, propyne, isobutane, propane)
 |
Complete the following activity.
Write the names of the hydrocarbons for the following structural formula.
(isobutylene, cyclohexane, propene, cyclohexene, cyclopentane, benzene, propyne, isobutane, propane)
 |
Considering MgCl2 as ionic compound and CH4 as covalent compound give any two differences between these two compounds.
Give an example for each of the following statement
A compound in which two Covalent bonds are formed.
Identify the incorrect statement and correct them.
- Like covalent compounds, coordinate compounds also contain charged particles (ions). So they are good conductors of electricity.
- Ionic bond is a weak bond when compared to Hydrogen bond.
- Ionic or electrovalent bonds are formed by mutual sharing of electrons between atoms.
- Loss of electrons is called Oxidation and gain of electron is called Reduction.
- The electrons which are not involved in bonding are called valence electrons.
Which of the following is the formula of Butanoic acid?
