Advertisements
Advertisements
प्रश्न
Explain the nature of the covalent bond using the bond formation in CH3Cl.
उत्तर
The structure of CH3Cl is given below:
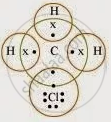
The molecule of chloromethane (CH₃Cl) is made up of three elements: carbon (Z = 6), hydrogen (Z = 1), and chlorine (Z = 17). Carbon has four valence electrons (2, 4), hydrogen has one (1), and chlorine has seven valence electrons (2, 8, 7). To achieve a full octet, carbon shares three of its valence electrons with three hydrogen atoms, and the fourth electron is shared with the chlorine atom.
APPEARS IN
संबंधित प्रश्न
What type of bonds are present in methane (CH4) and sodium chloride (NaCl)?
Give one example of a molecule containing a double covalent bond
Fill in the blanks in the following sentence:
In forming oxygen molecule, .............. electrons are shared by each atom of oxygen.
What type of bond is present in oxygen molecule?
Give two general properties of ionic compounds and two those of covalent compounds.
Using electron-dot diagrams which show only the outermost shell electrons, show how a molecule of oxygen, O2, is formed from two oxygen atoms. What name is given to this type of bonding? (At. No. of oxygen = 8)
Why is graphite a good conductor of electricity but diamond is a non-conductor of electricity?
The electronic configurations of three elements X, Y and Z are as follows:
| X | 2, 4 |
| Y | 2, 7 |
| Z | 2, 1 |
(a) Which two elements will combine to form an ionic compound?
(b) Which two elements will react to form a covalent compound?
Give reasons for your choice.
An element A has 4 valence electrons in its atom whereas element B has only one valence electron in its atom. The compound formed by A and B does not conduct electricity. What is the nature of chemical bond in the compound formed? Give its electron-dot structure.
Define a covalent bond.
Define a coordinate bond and give the conditions for its formation.
Explain the following term with example.
Hetero atom in a carbon compound
Molecular formula of Propane is C3H8 , write the structural formula of propane.
(a) Compound X consists of molecules.
Choose the letter corresponding to the correct answer from the choices (a), (b), (c) and (d) given below
The type of bonding in X will be
What is the term defined below?
A bond formed by a shared pair of electrons, each bonding atom contributing one electron to the pair.
Give reason as to why hydrogen chloride can be termed as a polar covalent compound.
Compare the compounds carbon tetrachloride and sodium chloride with regard to solubility in water and electrical conductivity.
An element L consists of molecules.
What type of bonding is present in the particles that make up L?
Draw an electron dot diagram to show the formation of the following compound.
Methane
Draw an electron dot diagram to show the formation of the following compound.
Magnesium chloride [ H=1, C=6, Mg=12, Cl=17].
What are the term defined below?
A bond formed by share pair of electrons, each bonding atom contributing one electron to the pair.
Draw the electron dot diagram and structure of nitrogen molecule.
Draw the electron dot diagram and structure of :
methane
What is the difference between a polar covalent compound and a non-polar covalent compound?
Explain the following:
Polar covalent compounds conduct electricity?
Taking MgCl2 as an electrovalent compound, CCl4 as a covalent compound, give four difference between electrovalent and covalent compounds
From the following hydrocarbon _______ is the cyclic hydrocarbon.
The number of electrons in the valence shell of a carbon atom is 4.
Write an Explanation.
Alkene
Complete the following activity.
Write the names of the hydrocarbons for the following structural formula.
(isobutylene, cyclohexane, propene, cyclohexene, cyclopentane, benzene, propyne, isobutane, propane)
 |
The number of pi-bonds present in benzoic acid molecule are ______.
Acetic acid was added to a solid X kept in a test tube. A colourless, odourless gas Y was evolved. The gas was passed through lime water, which turned milky. It was concluded that ______.
`"CH"_3 - "CH"_2 - "OH" overset("Alkaline""KMnO"_4 + "Heat")(->) "CH"_3 - "COOH"`
In the above given reaction, alkaline KMnO4 acts as
The table shows the electronic structures of four elements.
| Element | Electronic Structure |
| P | 2, 6 |
| Q | 2, 8, 1 |
| R | 2, 8, 7 |
| S | 2, 8, 8 |
- Identify which element(s) will form covalent bonds with carbon.
- “Carbon reacts with an element in the above table to form several compounds.” Give suitable reason.
In covalent bond formation, the sharing of ______ electrons takes place in their outermost shell.
"Carbon prefers to share its valence electrons with other atoms of carbon or with atoms of other elements rather than gaining or losing the valence electrons in order to attain noble gas configuration." Give reasons to justify this statement.
State the reasons, why carbon cannot
- Lose four electrons to form C4+ cation and
- Gain four electrons to form C4- anion.
How does carbon overcome this problem to form compounds?
