Advertisements
Advertisements
प्रश्न
Give the names and structural formulae of one saturated cyclic hydrocarbon and one unsaturated cyclic hydrocarbon.
उत्तर
Name of one saturated cyclic hydrocarbon is cyclohexane. It's molecular formula is C6H12.
Structure of cyclohexane is as follows:

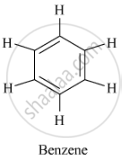
Name of one unsaturated cyclic hydrocarbon is benzene. It's molecular formula is C6H6.
Structure of benzene is as follows:
APPEARS IN
संबंधित प्रश्न
Draw the electron dot structures for ethanoic acid.
Write a short note on Catenation.
Write the electron-dot structures for ethene.
Write the electron-dot structures for ethyne.
An unsaturated hydrocarbon having a triple covalent bond has 50 hydrogen atoms in its molecule. The number of carbon atoms in its molecule will be:
(a) 24
(b) 25
(c) 26
(d) 28
You are given the following molecular formulae of some hydrocarbons:
C5H8; C7H14; C6H6; C5H10; C7H12; C6H12
Which two formulae represent unsaturated hydrocarbons having triple bonds?
Identify the unsaturated compounds from the following:
- Propane
- Propene
- Propyne
- Chloropropane
In which of the following compounds, — OH is the functional group?
Which among the following are unsaturated hydrocarbons?
- \[\ce{H3C - CH2 - CH2 - CH3}\]
- \[\ce{H3C - C ≡ C - CH3}\]
- \[\begin{array}{cc}
\ce{H3C - CH - CH3}\\
|\phantom{..}\\
\ce{CH3}\\
\end{array}\] - \[\begin{array}{cc}
\ce{H3C - C = CH2}\\
|\\
\phantom{...}\ce{CH3}\\
\end{array}\]
Name the reaction which is commonly used in the conversion of vegetable oils to fats. Explain the reaction involved in detail.
